1, 3-Cyclopentadlene reacts with Cycloheptatrienone to give the product shown. Tell what kind of reaction is Involved,...
Question:
1, 3-Cyclopentadlene reacts with Cycloheptatrienone to give the product shown. Tell what kind of reaction is Involved, and explain the observed result. Is the reaction suprafacial orantarafacial?
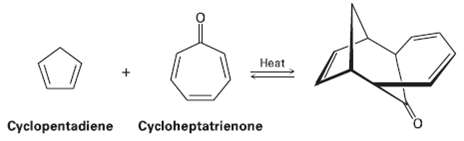
Transcribed Image Text:
Heat Cycloheptatrienone Cyclopentadiene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Diene HOMO Triene LUMO 4 heat The reaction of cyclopentadien...View the full answer

Answered By

Anurag Agrawal
I am a highly enthusiastic person who likes to explain concepts in simplified language. Be it in my job role as a manager of 4 people or when I used to take classes for specially able kids at our university. I did this continuously for 3 years and my god, that was so fulfilling. Sometimes I've skipped my own classes just to teach these kids and help them get their fair share of opportunities, which they would have missed out on. This was the key driver for me during that time. But since I've joined my job I wasn't able to make time for my passion of teaching due to hectic schedules. But now I've made a commitment to teach for at least an hour a day.
I am highly proficient in school level math and science and reasonably good for college level. In addition to this I am especially interested in courses related to finance and economics. In quest to learn I recently gave the CFA level 1 in Dec 19, hopefully I'll clear it. Finger's crossed :)
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give the product expected when benzoic acid reacts with each of the following reagents. (a) CH3I, K2CO3 (b) Concentrated HNO3, H2SO4 (c) PCl5 (d) P2O5, heat
-
What product is expected when cyclopentene reacts with iodobenzene in the presence of tri-ethlamine and a Pd(0) catalyst?
-
Give the product expected (if any) when butyraldehyde (butanal) reacts with each of the following reagents. (a) PhMgBr, then dilute H3O+ (b) LiAlH4 in ether, then H3O+ (c) Alkaline KMnO4, then H3O+...
-
Hooghly Products manufactures a silicone paste wax that goes through three processing departments- Cracking, Blending, and Packing. All raw materials are introduced at the start of work in the...
-
A corporate vision can be described narrowly or broadly. Google's website describes its mission/vision as organizing the world's information and making it universally accessible and useful. What does...
-
Construct the ATI curve for the sampling plan N = 1200, n = 50, c = 1. Suppose that the process average nonconforming rate is 3%. Explain the value of ATI for that level of nonconformance.
-
A six-cylinder, four-stroke petrol engine develops \(40 \mathrm{~kW}\) when running at \(3000 \mathrm{rpm}\). The volumetric efficiency at NTP is \(85 \%\). Indicated thermal efficiency is \(25 \%\)....
-
A severe winter ice storm has swept across North Carolina and Virginia followed by more than a foot of snow and frigid, single-digit temperatures. These weather conditions have resulted in numerous...
-
A baseball is thrown from the top of a building at a speed of 29 m / s with 54 of launch angle. The launch height is 20 m. Assume that air resistance is negligible. Find the baseball's final speed...
-
Journalize all these transactions into a general ledger. Transactions May 1 Purchased merchandise from Keys Wholesale on account for $735.30, Invoice No 91 Dated May 1, terms 2/10, n30 May 2...
-
What stereochemistry would you expect for the product of the DielsAlder reaction between (2E, 4E)-2, 4-hexadlene and ethylene? What stereochemistry would you expect if (2E, 4Z)-2, 4-hexadlene were...
-
Classify the following sigma tropic reaction by order [x, y], and tell whether it will proceed with suprafacial or antarafacial stereochemistry: -H H-
-
A gas has the equation of state \[\frac{p v_{m}}{\Re T}=1+\mathrm{A} p\left(T^{3}-9.75 T_{\mathrm{c}} T^{2}+9 T_{\mathrm{c}}^{2} T ight)+\mathrm{B} p^{2} T\] where \(\mathrm{A}\) and \(\mathrm{B}\)...
-
For this problem a 2d array of ints represents the value of each block in a city. Each element in the array is a city block. The value of a block could be negative indicating the block is a liability...
-
Thw case is from an Introduction to Finance Course at the Smith School of Business (COMM 121): WEIGHTED AVERAGE COST OF CAPITAL (FALL 2023): BOMBARDIER Shin Lee, a commerce student at the Smith...
-
------------------------------------------------------------------------------------------------------------ 1. Think of a client / website. A small to medium size company. Which website did you...
-
The advertising budgets of small stores in Los Angeles is normally distributed with a mean of $5,400 and a standard deviation of $2,100. What percentage of small stores in L.A. have an advertising...
-
From your analysis, you understand that the net margin for the organization has shown strong growth over the past year. What factor/s is/are driving this improved performance? What concerns do you...
-
Explain sustainability and the ESG movement in the field of management. What implications do they have for someone studying management?
-
An item of depreciable machinery was acquired on 1 July 2009 for $120,000 by cash It is expected to have a useful life of 10 years and zero salvage value On 1 July 2012, it was decided to revalue the...
-
An ATS (Alternative Trading System) might be defined as an exchange that does not operate as a self-regulatory body. True or false?
-
A certain natural product having [α]D + 40.3° was isolated. Two structures have been independently proposed for this compound. Which one do you think is more likely to be correct?...
-
One of the principal substances obtained from archaea (one of the oldest forms of life on earth) is derived from a 40-carbon diol. Given the fact that this diol is optically active, is it compound A...
-
(a) An aqueous solution containing 10 g of optically pure fructose was diluted to 500 mL with water and placed in a polarimeter tube 20 cm long. The measured rotation was -5.20. Calculate the...
-
Explain why leasing is an option for a company expansion. include, what leasing is and how it will benefit the company in it's expanding efforts. Also, how is capital or operating leasing recorded on...
-
Discuss the following statement: " A head of state signs a treaty on behalf of his country in excess of authority of his country, such treaty shal be void for inconsistency with domestic law of the...
-
A company is looking at new equipment with an installed cost of $415,329. This cost will be depreciated straight-line to zero over the project's 5-year life, at the end of which the equipment can be...

Study smarter with the SolutionInn App


