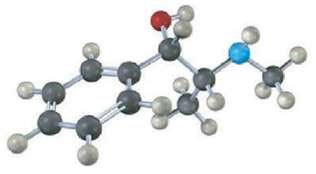
A Assign R or S configuration to each chirality center in pseudoephedrine, an over-the-counter decongestant found in
Question:
A Assign R or S configuration to each chirality center in pseudoephedrine, an over-the-counter decongestant found in cold remedies (blue =N).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
HO H sc ...View the full answer

Answered By

Muhammad Mahtab
everyone looks that their work be perfect. I have more than a five year experience as a lecture in reputable institution, national and international. I provide perfect solution in marketing, case study, finance problems, blog writing, article writing, business plans, strategic management, human resource, operation management, power point presentation and lot of clients need. Here is right mentor who help clients in their multi-disciplinary needs.
5.00+
3+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Charged particles in a vacuum are accelerated (In a device called a betatron) by the Electric Field E that necessarily accompanies a time-dependent magnetic ?eld B(t). Suppose that, in cylindrical...
-
Assign R or S configuration to each chirality center in the followingmolecules: (b) NH2 (a) . "C r "O
-
Assign R or S configuration to each chirality center in the followingmolecules: (a) (b) ,CH (c) , . CH CHCH
-
Your client has offered a 5-year, $1,000 par value bond with a 10 percent coupon. Interest on this bond is paid quarterly. 1) If your client is to earn a nominal rate of return of 12 percent,...
-
Discuss whether or not lower pressure is the result of fast-moving air, or fast-moving air is the result of lower pressure. Give one example supporting each point of view. (In physics, when two...
-
A thin, high-strength steel rule (E = 30 Ã 106 psi) having thickness t = 0.175 in. and length L = 48 in. is bent by couples M0 into a circular are subtending a central angle a = 40° (see...
-
What is the main concern that organizations have about the IoT?
-
A firm experienced the demand shown in the following table. a. Fill in the table by preparing forecasts based on a five-year moving average, a three-year moving average, and exponential smoothing...
-
7. Minimize the DFA A 1 0 0 0 B C 1 1 0 E 1 1
-
1. Factors in the marketing environment are broken into categories. Identify the opportunities and threats that emerged in BP's path toward introducing its GlutenWise menu, and sort them into...
-
Which, if any, of the following structures represent meso compounds? (Blue = N, yellow-green =Cl.) (b) (c)
-
Which of the following compounds are chiral? Draw them, and label the chirality centers. (a) 2, 4-Dimethylheptane (b) 5-Ethyl-3, 3-dimethylheptane (c) cis-l, 4-Dichlorocyclohexane (d) 4,...
-
The 20112012 tuitions (in thousands of dollars) of 4-year, private, not-for-profit universities and colleges are described by the density histogram in Fig. 98. Some of the tuitions are greater than...
-
You just deposited $35 in quarters and dimes. If you have the same number of quarters as dimes, how many coins total do you have?
-
There is a raffle for a 5970 prize, Out of 1,000 tickets, one ticket will win the prize, and the other tickcts will win nothing. If you have a ticket, what is the expected payoff?
-
Pam plans to make a square fence around a vacant lot. The perimeter of the fence should be at least 52 feet. What should be the measure of each side of the fence?
-
Alexis bought x boxes of cereal for $3 each, including tax, and paid with a $20 bill. Which expression represents the total amount of change she should receive?
-
The marching band is selling candy bars and fruit snacks for a fundraiser. Candy bars are sold for $2.50 and fruit snacks are $1.50. A total of 650 items were sold, bringing in $1,195. How many candy...
-
Ted is a forensic accountant at the law firm of Chambers, Bergweitz, and Rowe. He has worked there for 10 years and is called in on cases when his unique skills are needed by the different attorneys...
-
You are interested in investing and are considering a portfolio comprised of the following two stocks. Their estimated returns under varying market conditions are provided: (note: it is difficult to...
-
Which gas sample has the greatest volume at STP? a) 10.0 g Ar b) 10.0 g Kr c) 10.0 g Xe d) None of the above (They all have the same volume.)
-
Outline curved-arrow mechanisms for each of the known transformations shown in Fig. P22.85 that can be used to form three-membered rings. (Part (a) is an example of the Darzens glycidic ester...
-
Outline curved-arrow mechanisms for each of the known transformations shown in Fig. P22.85 that can be used to form three-membered rings. (Part (a) is an example of the Darzens glycidic ester...
-
Draw the "enol" isomers of the following compound. (The "enol" of a nitro compound is called aci-nitro compound, and the "enol" of an amide is called an imidic acid.) Ph-C NH2 benzamide
-
The market is expected to return 15 percent next year and the risk-free rate is 7 percent. What is the expected rate of return on a stock with a beta of 1.3? The covariance of the market's returns...
-
A stock's current price is 145.05. A put option with an exercise price of 120 and maturity of 3 months is currently priced at $ 28.83. What is the option's time value?
-
An investor put 60 percent of his money into a risky asset offering a 10 percent return with a standard deviation of return of 8 percent, and he put the balance of his risk-free asset offering 5...

Study smarter with the SolutionInn App


