How could each of the following compounds be converted to a. b. CH 3 CH 2 CH
Question:
How could each of the following compounds be converted to
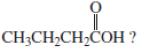
a.
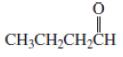
b. CH3CH2CH2CH2OH
c. CH3CH2CH2CH2Br
d. CH3CH2CH=CH2
Transcribed Image Text:
CH3CH2CH2COH?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
a b c d ...View the full answer

Answered By

Antony Mutonga
I am a professional educator and writer with exceptional skills in assisting bloggers and other specializations that necessitate a fantastic writer. One of the most significant parts of being the best is that I have provided excellent service to a large number of clients. With my exceptional abilities, I have amassed a large number of references, allowing me to continue working as a respected and admired writer. As a skilled content writer, I am also a reputable IT writer with the necessary talents to turn papers into exceptional results.
4.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify A-G: 1. CH3CCI AlCl3 . 1.03, -78 C
-
a. Give the products obtained from ozonolysis of each of the following compounds, followed by work-up under oxidizing conditions: b. What compound would form the following products upon reaction with...
-
Can carbon-nitrogen double and triple bonds be reduced by lithium aluminum hydride? Explain your answer.
-
Let (x, y) = x 4 2x 2 + y 2 6y. (a) Find the critical points of and use the Second Derivative Test to determine whether they are a local minima or a local maxima. (b) Find the minimum value of ...
-
What is meant by constant opportunity costs and increasing opportunity costs? Under what conditions will a country experience constant or increasing costs?
-
The shaft is made of bronze C86100 and has an elliptical cross section. If it is subjected to the torsional loading shown, determine the maximum shear stress within regions AB and BC, and the angle...
-
For the following set of scores, calculate the Pearson correlation coefficient and interpret the outcome. Budget Increase Number of Clients Served 7% 3% 11% 14% 5% 13% 7% 26% 2% 8%
-
a. Refer to the table below. What is the cumulative budgeted cost at the end of week 6? Amounts are in thousands of dollars. b. Below is a table of actual costs. What is the cumulative actual cost at...
-
Horticultural Products Inc. reported $616,709 profit in 2023 and declared preferred dividends of $48,700. The following changes in common shares outstanding occurred during the year. Jan. 1 48,000...
-
Below are incomplete financial statements for Bulldog, Inc. Required: Calculate the missing amounts. BULLDOG, INC. BULLDOG, INC. Statement of Stockholders' Equity Year ended Dec. 31, 2021 Income...
-
Give the products of the following reactions. Indicate whether each reaction is an oxidation or a reduction: a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. q. r. Na2Cr207 H2SOA CH3CH2CH2CH2CH2OH...
-
Identify the alkene that would give each of the following products upon ozonolysis followed by treatment with hydrogen peroxide: a. b. c. d. e. f. CH3CH2CH2COH CH3CCH CH3CCH2CH2CH2CH2CCH2CH3 OH HO +...
-
For each of the following reactions, (1) give the major 1,2- and 1,4-addition products and (2) indicate which is the kinetic product and which is the thermodynamic product: a. b. c. d. CH2 CH3 CH3...
-
Donald is resident and domiciled in the UK. He is not a Scottish taxpayer. He has the following income in tax year 2017-18: UK trading profits Income from UK property Income from foreign property...
-
During the year to 31 March 2018 , a company which qualifies as medium-sized spends 180,000 on research and development . This is qualifying expenditure under the R&D tax relief scheme. Explain the...
-
State the date (or dates) on which corporation tax is due for payment in relation to the following periods of account: (a) the year to 31 March 2018 (b) the six months to 30 November 2017 (c) the 21...
-
Sebastian is self-employed. He drives a ca r with an emission rating of 19 8 g/km and he charges the cost of all the petrol used to his business bank account . In the quarter to 31 July 2017 , he...
-
Tony died on 11 July 2017, leaving an estate valued at 9 00,000. None of the transfers made on his death were exempt from IHT. He had made the following transfers during his lifetime: 3 May 2009 1...
-
Sometimes a business entity will change its method of accounting for certain items. The change may be classified as a change in accounting principle, a change in accounting estimate, or a change in...
-
Fred Farmer needs to prepare a balance sheet for his bank. He spent the day getting the following information. Fred needs your help to build a balance sheet and evaluate it. The information was...
-
Compounds W and X are isomers; they have the molecular formula C9H8O. The IR spectrum of each compound shows a strong absorption band near 1715 cm-1. Oxidation of either compound with hot, basic...
-
The reaction of an aldehyde or ketone with a Grignard reagent is a nucleophilic addition to the carbon-oxygen double bond. (a) What is the nucleophile? (b) The magnesium portion of the Grignard...
-
Compounds Y and Z are isomers with the molecular formula C10H12O. The IR spectrum of each compound shows a strong absorption band near 1710 cm-1. The 1H NMR spectra of Y and Z are given in Figs. 16.4...
-
Convert the following numbers (Show the steps of your work): a. Ox23734 Binary: Decimal: b. Ob01100011 Hex: Decimal: c. 12345 Hex: Binary:
-
Find the total number of compounding periods and the interest rate per period for the investment. Nominal Term of Investment Interest (Annual) Rate (%) Compounded Compounding Periods Rate per Period...
-
A company applies overhead at a rate of 160% of direct labor cost. Actual overhead cost for the current period is $1,006,000, and direct labor cost is $590,000. Determine whether there is over- or...

Study smarter with the SolutionInn App


