(a) Estimate the activation energy for creep (i.e., Qc in Equation 8.20) for the S-590 alloy having...
Question:
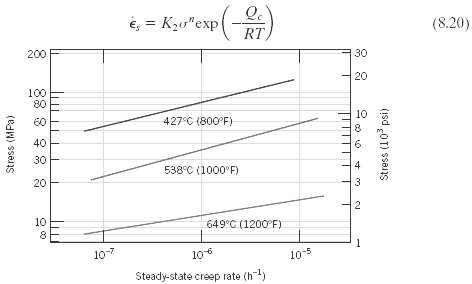
(a) Estimate the activation energy for creep (i.e., Qc in Equation 8.20) for the S-590 alloy having the steady-state creep behavior shown in figure. Use data taken at a stress level of 300MPa (43,500psi) and temperatures of 650°C and 730°C. Assume that the stress exponent n is independent of temperature.
(b) Estimate ?YS at 600°C (873 K) and300MPa.
Transcribed Image Text:
Q. é, = Kzu"exp(-) RT %3D (8.20) 200 30 20 100 80 10 60 427°C (800°F) 40 30 538°C (1000 F) 20 10 649°C (1200 F) 10-7 10 10-5 Steady-state creep rate (h) Stress (MPa) 3. Stress (10° psi)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a We are asked to estimate the activation energy for creep for the S590 alloy having the steadystate ...View the full answer

Answered By

Anum Naz
Lecturer and researcher with 10+ years of experience teaching courses in both undergraduate and postgraduate levels. Supervised 17 BA theses, 07 MA theses, and 1 Ph.D. dissertations. Edited and co-authored 2 monographs on contemporary trends in political thought. Published over articles in peer-reviewed journals.
4.80+
11+ Reviews
52+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
The activation energy for the reaction NO2(g) + CO(g) NO(g) + CO2(g) is 125 kJ/mol, and E for the reaction is 216 kJ/ mol. What is the activation energy for the reverse reaction. [NO(g) + CO2(g) ...
-
The activation energy for the reaction A2(g) + B2(g) 2AB(g) is 167 kJ/mol, and E for the reaction is 128 kJ/mol. What is the activation energy for the decomposition of AB?
-
The activation energy for a certain uncatalyzed biochemical reaction is 50.0 kJ/ mol. In the presence of a catalyst at 37oC, the rate constant for the reaction increases by a factor of 2.50 103 as...
-
Complete the questions in the table below.
-
On November 1 of year 0, Jaxon borrowed $50,000 from Bucksnort Savings and Loan for use in his business. In December, Jaxon paid interest of $4,500 relating to the 12-month period from November of...
-
Camco Manufacturers Inc., a publicly listed company, has two machines that are accounted for under the revaluation model. Technology in Camco's industry is fast-changing, causing the fair value of...
-
A centrifugal pump lifts water against a static head of 30 m of which suction lift is 3 m. The loss of head in suction pipe is 1 m and 5 m in delivery pipe. Speed of pump is 1440 rpm. The combined...
-
A large profitable corporation is considering two mutually exclusive capital investments: Alt A Alt B Initial cost $11.000 $33,000 Uniform annual benefit 3,000 9,000 End-of-depreciable-life 2,000...
-
Journalize the entry for the purchase of office supplies on February 13 for $2,300, paying $255 cash and the remainder on account. If an amount box does not require an entry, leave it blank. Feb. 13
-
The weight of a water tank open to the atmosphere is balanced by a counterweight, as shown in Fig. P13??41. There is a 4-cm hole at the bottom of the tank with a discharge coefficient of 0.90, and...
-
From Equation 8.19, if the logarithm of is plotted versus the logarithm of ?, then a straight line should result, the slope of which is the stress exponent n. Using Figure 8.31, determine the value...
-
Steady-state creep rate data are given below for nickel at 1000(C (1273 K): If it is known that the activation energy for creep is 272,000 J/mol, compute the steady-state creep rate at a temperature...
-
Which operators cannot be overloaded?
-
Why is management accounting required to take on an outward-looking role of contributing to business strategy by identifying, measuring and communicating financial information about a wider business...
-
A club consists of 16 men and 19 women. In how many ways can they choose a president, vice president, treasurer, and secretary, along with an advisory committee of six people (who are not officers)?
-
In Problems 12-15, match each formula in Column A with the type of financial problem in Column B. Column A 15. m = 1 P() - (1 + 2) -nt Column B Sinking fund
-
A speed control system of an engine is shown in Fig. P1.2. Determine: (a) Sensitivity of closed loop system to changes in engine gain \(k_{1}\) and tachometer feedback gain \(k_{2}\). (b) Steady...
-
Use a calculator to evaluate an ordinary annuity formula \[A=m\left[\frac{\left(1+\frac{r}{n}ight)^{n t}-1}{\frac{r}{n}}ight]\] for \(m, r\), and \(t\) (respectively) given in Problems 7-22. Assume...
-
Perfect Health Pharmacy allocates indirect expenses to its three departments on the basis of sales. For the year ended 30 June 2019, the following allocations were made. Assume that, during the year...
-
Is it a breach of fiduciary duty for a director of a real estate investment trust (REIT) negotiating a joint venture on behalf of the REIT with another director for the development of a portfolio of...
-
If you are designing a component that needs to be able to change temperature fairly quickly, should you use a material with a high or low specific heat? Explain your answer.
-
Copper (Cu) and platinum (Pt) both have the FCC crystal structure, and Cu forms a substitutional solid solution for concentrations up to approximately 6 wt% Cu at room temperature. Determine the...
-
Calculate the number of vacancies per cubic meter at 1000C for a metal that has an energy for vacancy formation of 1.22 eV/atom, a density of 6.25 g/cm3, and an atomic weight of 37.4 g/mol. (A) 1.49 ...
-
What is the composition, in atom percent, of an alloy that consists of 4.5 wt% Pb and 95.5 wt% Sn? The atomic weights for Pb and Sn are 207.19 g/mol and 118.71 g/mol, respectively. (A) 2.6 at% Pb and...
-
How do change interventions leverage advanced methodologies such as appreciative inquiry, design thinking, and complexity science to foster a holistic understanding of change dynamics, unlock latent...
-
what extent do change interventions leverage digital technologies, data analytics, and artificial intelligence to enhance change effectiveness, monitor change progress, and facilitate real-time...
-
Consider a project that requires spending of $15,000 immediately and generates $6,000 per year for the next 4 years (assume each $6,000 comes at the end of each year).What is the NPV of this project?...

Study smarter with the SolutionInn App


