A hydrocarbon vapor-liquid mixture at 250?F and 500 psia contains N2, H2S, CO2, and all the normal
Question:
A hydrocarbon vapor-liquid mixture at 250?F and 500 psia contains N2, H2S, CO2, and all the normal paraffins from methane to heptane. Use Figure to estimate the K-value of each component in the mixture. Which components will have a tendency to be present to a greater extent in the equilibriumvapor?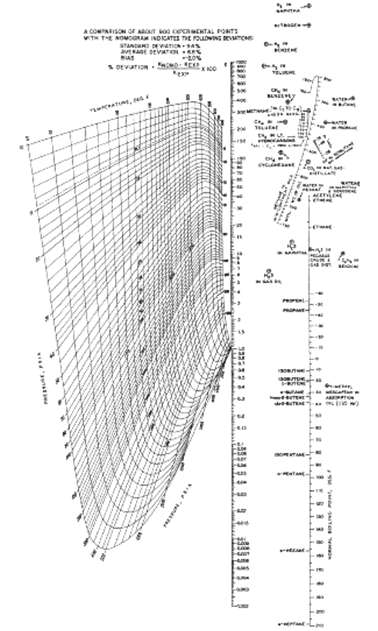
Transcribed Image Text:
A COAISON G t o cecaENTAL PONTE VT TE NOMCO INDICATES TE FouwINE Evmc STANNI DEVAnoSAS 13D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
If the Kvalue is 10 tendency is for vapor phase Using Fig ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
(a) What are the mole fractions of each component in a mixture of 15.08 g of O2, 8.17 g of N2, and 2.64 g of H2? (b) What is the partial pressure in atm of each component of this mixture if it is...
-
A 250 L rigid tank contains methane at 500 K, 1500 kPa. It is now cooled down to 300 K. Find the mass of methane and the heat transfer using a) ideal gas and b) the methane tables.
-
A 250 L rigid tank contains methane at 500 K, 1500 kPa. It is now cooled down to 300 K. Find the mass of methane and the heat transfer using a) ideal gas and b) the methane tables.
-
Consider Problem 13.28. The solvent MDEA becomes rich in acid gases. To recycle this solvent, it is first heated to 90C in exchanger E-2001 and then sent to the top stage of the stripper T-2002 as...
-
On June 30, $150,000 of five-year, 10% Plaza bonds are issued at $138,960 to yield a market interest rate of 12%. Interest is payable semi-annually each June 30 and December 31. (a) Record the...
-
According to the 2012 Value Line Investment Survey, the growth rate in dividends for JCPenney for the previous 10 years has been -7.5 percent. If investors feel this growth rate will continue, what...
-
A 5 -ft-long rod is used in a machine to transmit an axial compressive load of 3 kip. Determine its smallest diameter if it is pin connected at its ends and is made of a 2014-T6 aluminum alloy.
-
A local restaurateur who had been running a profitable business for many years recently purchased a three-way liquor license. This license gives the owner the legal right to sell beer, wine, and...
-
List and discuss three of the major issues in Homeland Security. If you were you were to rank these in order with the most serious being number one, what would that ranking look like? For each...
-
You have been employed as a consultant to Golf R Us, Inc. The company has been having problems with account receivables and they want to use various dunning methods to better their receivables. But...
-
Isopropanol, containing 13 wt% water, can be dehydrated to obtain almost pure isopropanol at a 90% recovery by azeotropic distillation with benzene. When condensed, the overhead vapor from the column...
-
Acetone, a valuable solvent, can be recovered from air by absorption in water or by adsorption on activated carbon. If absorption is used, the conditions for the streams entering and leaving are as...
-
Janet is planning to open a small car-wash operation, and she must decide how much space to provide for waiting cars. Janet estimates that customers would arrive randomly (i.e., a Poisson input...
-
Which is bigger, the exchange-traded market or the OTC market?
-
Explain how hedging using futures can lead to cash-flow problems such as those experienced by Metallgesellschaft.
-
How do the margin requirements for a small retail trader differ from those for a member of the exchange clearinghouse?
-
What is the difference between the clean price and the dirty price of a bond?
-
How is par yield defined?
-
Numerically calculate a histogram of the distribution of eigenvalues of \(\Gamma \times \Gamma\) GOE random matrices for moderate values of \(\Gamma\), and show that the distribution \(P(\lambda)\)...
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
An experimental station wishes to test whether a growth hormone will increase the yield of wheat above the average value of 100 units per plot produced under currently standard conditions. Twelve...
-
What is a reasonable value for the optimal absorption factor when designing an absorber? Does that same value apply to the optimal stripping factor when designing a stripper?
-
What is the difference between an operating line and an equilibrium curve?
-
For a given recovery of a key component in an absorber or stripper, does a minimum absorbent or stripping agent flow rate exist for a tower or column with an infinite number of equilibrium stages?
-
How should an employer considered the personal needs and development of employees when creating messaging? Supported your response by using a scholarly source(s) .
-
You are ordering a new home theater system that consists of a TV, surround sound system, and DVD player. You can choose from 7 different TVs, 6 types of surround sound systems, and 6 types of DVD...
-
A tool manufacturing company, Mississippi Tools Inc., went bankrupt and the Court appointed a Licensed Insolvency Trustee to liquidate the estate of the bankrupt company. The Trustee has taken the...

Study smarter with the SolutionInn App


