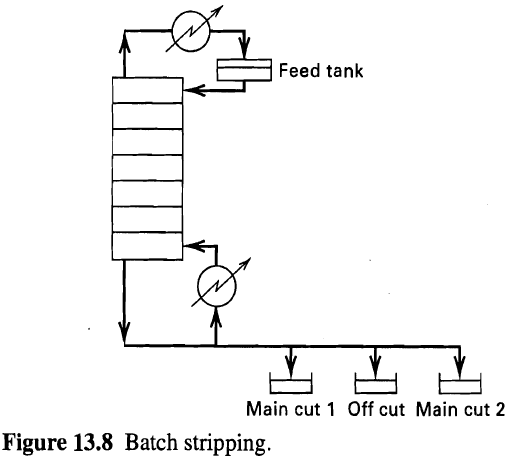
A three-theoretical-stage batch stripper (one stage is the reboiler) is charged to the feed tank (see figure)
Question:
A three-theoretical-stage batch stripper (one stage is the reboiler) is charged to the feed tank (see figure) with 100 kmol of 10 mol% n-hexane in n-octane mix. The boilup rate is 30 kmol/h. If a constant boilup ratio (V/L) of 0.5 is used, determine the instantaneous-bottoms composition and the composition of the accumulated bottoms product at the end of 2 h of operation.
Transcribed Image Text:
Feed tank Main cut 1 Off cut Main cut 2 Figure 13.8 Batch stripping.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
Subject Batch stripping of a mixture of normal hexane H and normal octane O Given Charge of 100 kmol of 10 mol H and 90 mol O Boilup up rate V 30 kmol...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
One kmol of octane (C8H18) is burned with air that contains 21 kmol of O2. Assuming the products contain only 1) CO2, 2) H2O, 3) O2 and 4) N2, determine the. (a) Mole number of each gas in the...
-
One compartment of an insulated rigid tank contains 2 kmol of CO2 at 20oC and 150 kPa while the other compartment contains 5 kmol of H2 gas at 35oC and 300 kPa. Now the partition between the two...
-
A rigid tank contains 4 kmol O2 and 5 kmol of CO2 gases at 18oC, 100 kPa. Determine (a) the volume () of the tank.
-
Perform computational experiments to verify that the average path length in a ring graph on \(V\) vertices is \(\sim 1 / 4 V\). Then, repeat these experiments, but add one random edge to the ring...
-
On its acquisition-date consolidated balance sheet, what amount should TruData report as common stock? a. $70,000 b. $300,000 c. $350,000 d. $370,000 On July 1, TruData Company issues 10,000 shares...
-
A merchant bought an article for $7.92. How much did the article sell for if he sold it at an increase of 83 1/3%?
-
A-Mobile Wireless needed additional capital to) expand, so the business incorporated. The charter from the state of Georgia authorizes A-Mobile to) issue \(50,(0) 0)\) shares of \(6 \%, \$ 100\)-par...
-
Alina Sadofsky, who operates a small grocery store, has established the following policies with regard to the checkout cashiers: 1. Each cashier has his or her own cash drawer, to which no one else...
-
A 3 0 - year semi - annual bond was issued 2 0 years ago. Coupon rate = 5 % . a. What is the interest rate if the current price = $ 8 8 0 ? b. What is the interest rate if the current price = $ 1 , 1...
-
Start of Payroll Project 7-3a October 9, 20-- No. 1 The first payroll in October covered the two workweeks that ended on September 26 and October 3. This payroll transaction has been entered for you...
-
Develop a procedure similar to that of Section 13.2 to calculate a binary batch stripping operation using the equipment arrangement of figure 13.8. Feed tank Main cut 1 Off cut Main cut 2 Figure 13.8...
-
Develop a procedure similar to that of Section 13.2 to calculate a complex, binary, batch-distillation operation using the equipment arrangement of figure.
-
Using the following accounts, prepare a single-step income statement at year end, May 31, 2014: Cost of Goods Sold, $1,680; General Expenses, $900; Interest Expense, $420; Interest Income, $180; Net...
-
Using the output in Figure 1, write a program that uses the Encapsulation concept where: i. Make the instance variables private. ii. Have getter and setter methods in the class to set and get the...
-
PERFORMANCE TASK IN PCOM CREATE AN ARGUMENTATIVE ESSAY ABOUT: (MAKE A CATCHY TITLE ABOUT YOUR ESSAY) IS ANIMAL TESTING MORAL? In making argumentative essay Make sure to: 1. Make a claim 2. Develop...
-
Determine the support reactions at base O of the street light shown. Note that a wind is blowing in the positive y direction which puts and additional 20 lb load on each of B and C. B 100 lb 25' C...
-
1 Find the compound amount and compound interest if $4000 is invested for eight years and interest is compounded continuously at the annual rate 6%. The compound amount will be $ (Round to the...
-
2. Usually, Rebecca's bakery posts petty cash transactions and replonishes the account, which holds $350, every week. Rebecca's accountani moxed her petty cash receipts for the week of February 1-7...
-
The substance producing a lot of heat on burning is called as: (a) Oxidizing agent (b) Biogas (c) Biomass (d) Fuel
-
What are technical skills At what level are they most important and why?
-
What are derivatives? Define forward, futures, and option agreements. What are the underlying instruments in these agreements?
-
Hydrocarbon fuels can be produced from methanol by reactions such as the following, which yieklds 1-hexene: 6CH3OH(g) ( C6H12(g) + 6H2O(g) Compare the standard heat of combustion at 25(C of OCThOHigl...
-
Calculate the theoretical flame temperature when ethylene at 25(C is burned with: (a) The theoretical amount of air at 25(C. (b) 25% excess air at 25(C. (c) 50% excess air at 25(C. (d) 100% excess...
-
For steady Bow through a heat exchanger at approximately atmospheric pressure, what is the final temperature, (a) When heat in the amount of 800 kJ is added to 10 mol of ethylene initially at 200(C?...
-
Copr. Goedl UFacture is considering a new stamping machine. The machine costs $225,000. new machine can be used to generate $66,000 in annual revenue. Cash operation expens are estimated to be...
-
Titanic Corporation's net income for the year ended December 31, 2022, is $380,000. On June 30, 2022, a $0.75 per-share cash dividend was declared for all common shareholders. Outstanding at the time...
-
. Develop a pricing strategy for your product/service. Consider factors such as cost, competitor pricing, and perceived value. Psychological Pricing (300 words): Discuss how psychological factors...

Study smarter with the SolutionInn App


