A total of 6,000 lb/h of a liquid solution of 40 wt% benzene in naphthalene at 50?C
Question:
A total of 6,000 lb/h of a liquid solution of 40 wt% benzene in naphthalene at 50?C is cooled to 15oC. Assuming that equilibrium is achieved, use Figure to determine the amount of crystals formed, and the flow rate and composition of the mother liquor. Are the crystals benzene or naphthalene?
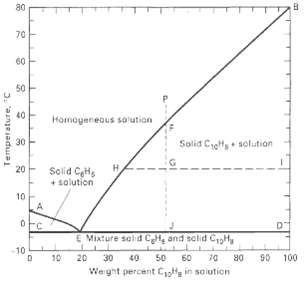
Transcribed Image Text:
a0 70 60 50 40 Hamogeneous so'ution Solid Cett, • solution 30 Solid CeHs + solution 20 10 E Mixture sald CH, and sclid CHy 10 S0 100 20 30 50 60 10 40 80 Weight percent Coa in saiution Temperature, "C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The feed is 0606000 3600 lbh of naphthalene and 2400 lbh of benzene In the figure bel...View the full answer

Answered By

Ashish Jaiswal
I have completed B.Sc in mathematics and Master in Computer Science.
4.90+
20+ Reviews
39+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
A liquid containing 60 mol% toluene and 40 mol% benzene is continuously distilled in a single-equilibrium-stage unit at atmospheric pressure. What percent of benzene in the feed leaves in the vapor...
-
Benzene flowing at 12.5 kg/s is to be cooled continuously from 80?C to 54?C by 10 kg/s of water available at 15.5?C. Using Table 8.5, estimate the surface area required for(a) Cross-flow with six...
-
solution of Na2SO4. Determine the composition and flow rate of the product if the flow rate of Na2SO4 is 1275 kg/hr, and the ratio of the flow rate of the H2O to the product solution is 0.83.
-
Describe what happens if you put the double quotes in the print statement of HelloWorld.java on different lines, as in this code fragment: System.out.println("Hello, World");
-
On May 31, O'Hearn Limited had a cash balance per books of $13,400. The bank statement from Community Bank on that date showed a balance of $15,230. A comparison of the bank statement with the...
-
Pam Corporation purchased 75 percent of the outstanding voting stock of Sun Corporation for $4,800,000 on January 1, 2016. Sun's stockholders' equity on this date consisted of the following (in...
-
How can a test be reliable and not valid? And why is a test not valid unless it is reliable?
-
Billings Company has the following information available for September 2017. Unit selling price of video game consoles $ 400 Unit variable costs ............. $ 280 Total fixed costs...
-
what are the characteristics of a complex trust? An estate with one beneficiary generates income of $10,000 in one year. That same year it distributes $15,000 to the beneficiary in the closing of the...
-
for the system shown below, design the complete oc protection using the idmt relays. thus, decide the ct ratios, the plug settings and the tms at all locations. 115 A 1500 A 6000 A 80 A 1000 A 77 A...
-
Repeat Exercise 4.53 if the residence time is only sufficient to leach 80% of the carbonate.
-
Repeat Example 4.10, except determine the temperature necessary to crystallize 80% of the naphthalene.
-
Palladium crystallizes with a face-centered cubic structure. It has a density of 12.0 g/cm 3 , a radius of 138 pm, and a molar mass of 106.42 g/mol. Use these data to calculate Avogadros number.
-
Repeat Exercise 9.9 for the lowpass analysis filter equal to \[H_{0}(z)=z^{-3}-z^{-2}+z^{-1}+1 .\] Exercise 9.9 Consider a perfect reconstruction filter bank which satisfies the QMF condition,...
-
A large manufacturer purchases an identical component from three independent suppliers that differ in unit price and quantity supplied. The relevant data for 2009 and 2011 are given here. a. Compute...
-
Verify that the set of matrices (5.14) is closed under ordinary matrix multiplication. Data from Eq. 5.14 T(oc)= = 629 > - (+19) TOO) = (721) TO) = ( ). T(oa)= T(b) TO) -(11) T(4-(11) TO=(9) T(C3)= =
-
A major manufacturing company reports the quantity and product value information for 2007 and 2011 in the table that follows. Compute a weighted aggregate quantity index for the data. Comment on what...
-
Compute the price relatives for the four stocks making up the Boran index in exercise 20. Use the weighted aggregates of price relatives to compute the January 2011 and March 2011 Boran indexes. Data...
-
In fruit flies, dosage compensation is achieved by a. X-chromosome inactivation. b. doubling the expression of genes on the single X chromosome in the male. c. halving the expression of genes on each...
-
Modify the counter from Exercise 5.44 such that the counter will either increment by 4 or load a new 32-bit value, D, on each clock edge, depending on a control signal Load. When Load = 1, the...
-
Outline the direct and indirect costs to an organization of turnover.
-
What is differential (Rayleigh) distillation? How does it differ from batch rectification?
-
How does batch distillation differ from continuous distillation?
-
The upper column of an air-separation system of the type shown in Exercise 7.40 contains 48 sieve trays and operates at a nominal pressure of 131.7 kPa. A feed at 80 K and 131.7 kPa enters the top...
-
Find a linear equation for the line that passes through (7,5) and (3,17)
-
Your firm has just been retained by Harley Quinn (38 years old) for a divorce and custody case. She and John Joker (42 Years Old) have been married for 10 years. She is currently residing in the...
-
Explain the difference between a "SELECT" and an "INSERT" SQL statement.
Statistical Methods For Data Analysis In Particle Physics 1st Edition - ISBN: 3319201751 - Free Book

Study smarter with the SolutionInn App


