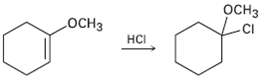
Addition of HC1 to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclo- hexane as the sole product. Use resonance structures to explain
Question:
Addition of HC1 to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclo- hexane as the sole product. Use resonance structures to explain why none of the other regioisomer isformed.
Transcribed Image Text:
оснз оСнз HCI CI CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
ficos OCH 3 H OCH3 Conjugation with the oxygen lone pair electrons makes the double bond more nucl...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Addition of HC1 to 1, 2-dimethylcyclohexene yields a mixture of two products. Show the stereochemistry of each, and explain why a mixture is formed.
-
Use resonance structures to help you identify all sites of low electron density (δ+) in the following compound:
-
Use resonance formulas to explain why polyacetylene has delocalized molecular orbitals extending over the length of the molecule, whereas the following molecule does not. HHHHH
-
Pipestone Ltd. (Pipestone) uses an aging schedule to estimate the amount of receivables that won't be collected. Pipestone allows its customers up to 60 days to pay amounts owed. Any receivable...
-
Why can't car dealerships simply keep paying based on past systems?
-
Obtain the Fourier series expansion for the waveform shown in Fig. 17.49. z(t)
-
Stabilized effluent from a hydrogenation unit as given below is to be separated by ordinary distillation into five relatively pure products. Four distillation columns will be required. According to...
-
Go to Marriotts website (www.marriott.com) and peruse the different types of lodging brands offered. Select any three and based on the discussion of differentiation in the chapter, identify the...
-
Explain the principles and applications of root cause analysis (RCA) in quality control, examining methods such as the "5 Whys," fishbone diagrams, and fault tree analysis for identifying underlying...
-
A fraction nonconforming control chart with n = 400 has the following parameters: UCL = 0.0962; Center line = 0.0500; LCL = 0.0038. (a) Find the width of the control limits in standard deviation...
-
Myrcene, C10H16, is found in oil of bay leaves and is isomeric with ?-Ocimene (Problem 14.48). It has an ultraviolet absorption at 226 nm and can be catalytically hydrogenated to yield 2,...
-
Hydrocarbon A, C 10 H 14 , has a UV absorption at ? max = 236 nm and gives hydrocarbon B, C 10 H 18 , on catalytic hydrogenation. Ozonolysis of A followed by zinc/acetic acid treatment yields the...
-
Students are often asked to write term papers containing a certain number of words. Counting words in a long paper is a tedious task, but the computer can help. Write a program that counts the number...
-
The return on the Super Traders share portfolio over the past six years are reported as follows: Year / Return 2 0 1 8 - 1 1 . 5 % 2 0 1 9 - 5 . 2 % 2 0 2 0 - ( - 1 . 5 % ) 2 0 2 1 - 1 6 . 5 % 2 0 2...
-
The diagram below shows the sound spectrum of a piano playing the A3 note. Relative 1.0 Amplitude 05 00 Piano 1000 2000 Frequency (Hz) 3000 a. Which line would represent the fundamental frequency? b....
-
The dot and metal. For a single-level quantum dot in contact with a metal, assuming that is independent of wo and that ||
-
Tell an appropriate lawyer joke that cannot be found online. Make it the best.
-
1. When F. Scott Fitzgerald was just a teenager, he realized that he had a talent for writing. He decided to use this talent to write plays, which he produced at his school. What was his main...
-
Using real-world examples, evaluate the effectiveness of demand-side policies in reducing unemployment.
-
One hundred pounds of water at atmospheric pressure are heated from 60F to 200F. What is the enthalpy change? The internal energy change? Why is the difference between the internal energy change and...
-
A solution contains 22.4 g glucose (C 6 H 12 O 6 ) dissolved in 0.500 L of water. What is the molality of the solution? (Assume a density of 1.00 g/mL for water.) a) 0.238 m b) 44.8 m c) 0.249 m d)...
-
Which of the following compounds would give a positive Tollens test? (Remember that the Tollens test involves mild basic aqueous conditions.) (a) CH3CH2CH2COCH3 (b) CH3CH2CH2CH2CHO (c)...
-
Solving the following road-map problem depends on determining the structure of A, the key intermediate. Give structures for compounds A through K. hept-1-yne SOCI CH3CuLi (2) (CHj,S (2) Ho HCN (2)...
-
The UV spectrum of an unknown compound shows values of λmax at 225 nm (ε = 10,000) and at 318 nm 1e = 402. The mass spectrum shows a molecular ion at m/z 96 and a prominent...
-
Using Fourier Transforms, solve the following integral Equation for ((t) f (t) - ( ( ( t -u) ue - 2 u H (u) du = foo_flt -4t e H(t)
-
Q1: what is the net operating income under the variable and absorption cost system. A: ACER company Product sales: 1,000 units at $10 each Variable manufacturing costs: $5.50 per unit Fixed...
-
Solve for 2 and graph the solution on the number line below. 245x+9 or 5x +9 69

Study smarter with the SolutionInn App


