Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield
Question:
Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:
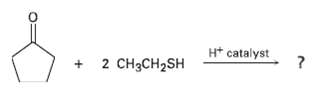
Transcribed Image Text:
H* catalyst 2 CH3CH2SH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
The same series of steps used to form an acetal is followed in thi...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the product of the following reaction. M 1) LIAID, 2) H20 Me
-
Predict the product of the following reaction. Br NaOCH3, heat -NO2
-
Predict the product of the reaction if CH3-S-CH2CH2CH2CH2-Br is heated in a polar organic solvent, such as methanol. Similarly, what would be the product for hexyl bromide in methanol? (In hexyl...
-
1. Review the six (6) goals of performance based acquisition(s); choose two and discuss how your choices can be implemented for effective management of contracts, using an actual or theoretical...
-
1. What factors of group cohesiveness were present in this team? 2. What should Joe do now to ensure the team's effectiveness and ultimate success? Use the Model of Team Effectiveness (PPT 15-9) to...
-
Factor the square of the binomial. (a) x2 + 12x + 36 (b) y2 - 6y + 9 (c) 9z2 - 12z + 4
-
Who is accountable for performing verification?
-
The following are the major balance sheet classifications: Current assets (CA).......... Current liabilities (CL) Long-term investments (LTI)....... Long-term liabilities (LTL) Property, plant, and...
-
Five years ago, State X bought Railroad (RR), which was in bankruptcy and about to be liquidated. RR has always been the largest rail carrier in State X, presently carrying 70% of its rail freight....
-
Bonds 1. Municipal Bonds - Municipal bonds are haircut per Exhibit 1 based on both their time to maturity and scheduled maturity at date of issue. 2. Corporate Bonds - Corporate bonds are haircut...
-
6-Methyl-5-hepten-2-one is a constituent of lemongrass oil. How could you synthesize this substance from methyl4-oxopentanoate? CH3CH2CH2OCH3 Methyl 4-oxopentanoate
-
Ketones react with dimethyl-sulfonium methylide to yield epoxides. Suggest a mechanism (or the reaction. HICHJ)2 DMSO solvent (CH3)2S Dimethylsulfonium methylide
-
How are human population growth and affluence related to natural resource depletion?
-
Kirito is planning to invest his retirement benefit amounting to Php 1,500,000. He was given 3 options on where to invest his money. Help Kirito decide on which bank/ cooperative will yield him the...
-
Cherokee Incorporated is a merchandiser that provided the following information: Amount Number of units sold 1 1 , 0 0 0 Selling price per unit $ 1 6 Variable selling expense per unit $ 2 Variable...
-
What is Debra's net worth if her assets total $40000, her gross income is $165000, her student loan debt is $70000 (she has no other debts), and her annual expenses (including taxes) total $88000?
-
Greenwood Company manufactures two products-13,000 units of Product Y and 5,000 units of Product Z. The company uses a plantwide overhead rate based on direct labor-hours. It is considering...
-
Reddy Company is a merchandising firm. Next month the company expects to sell 500 units. The following data describe the company's revenue and cost structure: Selling price per unit. $60 Sales...
-
While we introduced the variational method and the power method both as a way to approximate the ground state of some system, they both can be used to approximate excited states as well, with...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Explain why it is not necessary to find the Lewis structure with the smallest formal charges to make a successful prediction of molecular geometry in the VSEPR theory. For example, write Lewis...
-
Write the mechanistic steps in the cyclization of ethyl phenylacetoacetate (ethyl 3-oxo-4-phenylbutanoate) in concentrated sulfuric acid to form naphthoresorcinol (1,3-naphthalenediol).
-
When an aldehyde or a ketone is condensed with ethyl a-chloroacetate in the presence of sodium ethoxide, the product is an α,β-epoxy ester called a glycidic ester. The...
-
The Perkin condensation is an aldol-type condensation in which an aromatic aldehyde (ArCHO) reacts with a carboxylic acid anhydride, (RCH2CO)2O, to give an a,b-unsaturated acid (ArCH "CRCO2H). The...
-
Estimate the hydrogen consumption required to completely remove the sulfur from a hydrotreater feedstock and to reduce the nitrogen content of the product to 15 ppm by weight. The 48.5 API naphtha...
-
A firm has 36.30 million shares of common stock outstanding at a price of $32.81 per share. The firm also has 492000.00 bonds outstanding with a current price of $1,089.00. The outstanding bonds have...
-
Commwest Bank owns $300 million of variable-rate notes yielding BBR plus 4 per cent. These loans are financed by $300 million of fixed-rate bonds costing 5 per cent.Northside Building Societyhas $300...

Study smarter with the SolutionInn App


