Among the numerous examples of SN1 reactions that occur with incomplete racemization is one reported by Winstein
Question:
Among the numerous examples of SN1 reactions that occur with incomplete racemization is one reported by Winstein in 1952? The optically pure tosylate of 2, 2-dimethy-1-phenyl-1-propanol ([?] D = ? 30.3o) was heated in acetic acid to yield the corresponding acetate ([?] D = + 5.3o). If complete inversion had occurred, the optically pure acetate would have had [?] D = ? 53.6o. What percentage racemization and what percentage inversion occurred in this reaction?
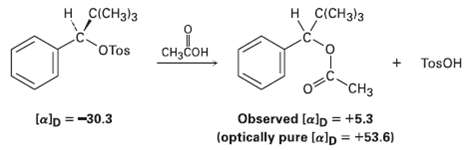
Transcribed Image Text:
H CICH3)3 н СICH3)з OTOS сHзCон TosOH "CHз Observed [alp = +5.3 (optically pure [alp = +53.6) lalp =-30.3 %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
If reaction had proceeded with complete inversion the product ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Would you expect pure acetic acid to be a strongly hydrogenbonded substance? How do the melting and boiling points of the substance (16.7C and 118C) support your answer?
-
Optically pure (S)-(+)-2-butanol was converted to its methanesulfonate ester according to the reaction shown. (a) Write the Fischer projection of the sec-butyl methanesulfonate formed in this...
-
Reaction of benzanilide with chlorine in acetic acid yields a mixture of two monochloro derivatives formed by electrophilic aromatic substitution. Suggest reasonable structures for these two isomers....
-
Suppose that a typical taxpayer has a marginal personal income tax rate of 35 percent. The nominal interest rate is 13 percent, and the expected inflation rate is 8 percent. a. What is the real...
-
Aside from the legal requirements, how should the principles of justice shape any efforts by employers to monitor employees' personal use of social media?
-
How many electrons are in the valence d orbitals in these transition-metal ions? (a) Co3+, (b) Cu+, (c) Cd2+, (d) Os3+,.
-
Explain how an auditor determines whether all recorded payables actually represent bona fide obligations of the entity.
-
Rehab Health Care, LLC, incurred the following service-related activity costs for the month: Total sales ................$42,000 Customer complaint processing ....... 1,200 Employee training...
-
What force will you need to push 200kg box with along a rough surface to get it to move a constant speed if the force of friction is 300N?
-
1. Using the spreadsheet model from Case 2.1 as a starting point, use Solver to find the optimal set of projects to approve. The solution should maximize the total NPV from the approved projects, and...
-
What product(s) would you expect from reaction of (S)-3-chloro-3-methyloctane with acetic acid? Show the stereo chemistry of both reactant and product.
-
Assign configuration to the following substrate, and show the stereo chemistry and identity of the product you would obtain by SN1 reaction with water (reddish brown =Br):
-
Voters in the State of California decide that the tobacco industry is having too great an impact on the outcome of local referenda limiting smoking in public places. To curb the influence of that...
-
Alex, Incorporated, buys 30 percent of Steinbart Company on January 1, 2023, for $1,098,000. The equity method of accounting is to be used. Steinbart's net assets on that date were $3.30 million. Any...
-
In January, Tongo, Incorporated, a branding consultant, had the following transactions. Indicate the accounts, amounts, and direction of the effects on the accounting equation under the accrual...
-
A high-speed rail line is proposed to connect two large cities 200 miles apart. The "constant speed" is expected to be 110 mph, with acceleration and deceleration rates at 3 ft/sec/sec.. A. How much...
-
What is the likely rationale behind excluding persons convicted for certain offences from possessing guns? A belief that convicted persons are more likely to commit further crimes with firearms than...
-
ERP and CRM in Business Mostlarge businesses rely on many complex processes to keep them running smoothly. Two large areas that impact large areas of business and IT are ERP and CRM. Respond to the...
-
Frame \(S^{\prime}\) moves relative to frame \(S\) as shown in Figure Q27.2. a. A ball is at rest in frame \(S^{\prime}\). What are the speed and direction of the ball in frame \(\mathrm{S}\) ? b. A...
-
A summary of changes in Pen Corporation's Investment in Sam account from January 1, 2011, to December 31, 2013, follows (in thousands): ADDITIONAL INFORMATION 1. Pen acquired its 80 percent interest...
-
Coal can be used to generate hydrogen gas (a potential fuel) by the endothermic reaction: If this reaction mixture is at equilibrium, predict whether each disturbance will result in the formation of...
-
Section 17-12 showed how nucleophilic aromatic substitution can give aryl amines if there is a strong electron-withdrawing group ortho or para to the site of substitution. Consider the following...
-
The following spectra for A and B correspond to two structural isomers. The NMR singlet at (1.16 in spectrum A disappears when the sample is shaken with D2O.The singlet at (0.6 ppm in the spectrum of...
-
A drug user responded to an ad placed by a DEA informant in a drug-culture magazine. He later flew from Colorado to Maryland, where he bought some 1-phenyl-2-propanone (P2P) from the informant. The...
-
Common law vs civil law system Judge made law Rule-based adjudication Fact-specific adjudication Jurisdiction Judicial hierarchy Rules vs standards/principles Ruel of law Constitutionalism Textualism...
-
The operating system is the physical environment where applications run. Any vulnerability in the operating system could compromise the security of one or more applications. Threats can come through...
-
Halftime in the football game on a Sunday afternoon means time for food at Charlie Masterson's house, and Masterson is ready for his favorite, a tuna sandwich and potato chips. Particularly hungry...

Study smarter with the SolutionInn App


