An alternative mining plan is to develop two levels, below the upper A Level and mine only
Question:
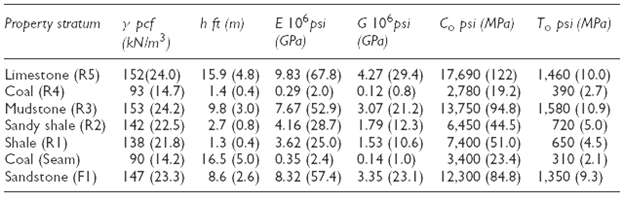
An alternative mining plan is to develop two levels, below the upper A Level and mine only the high grade. The B Level would be in the 13.1 m of high grade, while the C level would be in the lower 6.10 m of high grade in the 31.1 m column shown in the detailed geologic column. Determine the maximum possible roof span for the BLevel.
Transcribed Image Text:
h ft (m) Co psi (MPa) To psi (MPa) Property stratum E 10°psi G 10ʻpsi (GPa) y pef (kNim) (GPa) Limestone (R5) Coal (R4) Mudstone (R3) Sandy shale (R2) Shale (RI) Coal (Seam) Sandstone (FI) 152(24.0) 93 (14.7) 153 (24.2) 142 (22.5) 138 (21.8) 90 (14.2) 147 (23.3) 15.9 (4.8) 9.83 (67.8) 4.27 (29.4) 0.29 (2.0) 1.460 (10.0) 17,690 (122) 390 (2.7) 2,780 (19.2) 13,750 (94.8) 1,580 (10.9) 720 (5.0) 6,450 (44.5) 650 (4.5) 7,400 (51.0) 310 (2.1) 3,400 (23.4) 12,300 (84.8) 1,350 (9.3) 0.12 (0.8) 1.4 (0.4) 9.8 (3.0) 7.67 (52.9) 3.07 (21.2) 2.7 (0.8) 4.16 (28.7) 1.79 (12.3) 1.3 (0.4) 3.62 (25.0) 1.53 (10.6) 16.5 (5.0) 0.35 (2.4) 8.6 (2.6) 8.32 (57.4) 3.35 (23.1) 0.14 (1.0)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (16 reviews)
Given New mining plan Problem 37 183 335 m3 427 131 m 76 m 610 m Bonnete...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Geological Engineering questions
-
With reference to Problem 9, determine the maximum possible roof span, given that all roof rock must have a safety factor greater thanone. To (102) psi (MPa) Co (103, psi (MPa) E (106) psi (GPa) Rock...
-
For Problem 15-16, develop upper and lower con-trol limits for the range. Do these samples indicate that the process is in control? In Problem 15-16, Colonel Electric is a large company that produces...
-
For Problem 16-16, develop upper and lower control limits for the range. Do these samples indicate that the process is in control?
-
Average rates of return on Treasury bills, government bonds, and common stocks, 1900-2020. Average Annual Average Premium (Extra Rate of Return return versus Treasury (8) bills) (%) Portfolio...
-
A television is tuned to a station broadcasting at a frequency of 2.04 108 Hz. For best reception, the rabbit-ear antenna used by the TV should be adjusted to have a tip-to-tip length equal to half...
-
Is e-business likely to be more beneficial in the early part or the mature part of a products life cycle? Why?
-
Identify at least three grounds for an involuntary dismissal of an action.
-
(a) Mylar Corporation has collected the following information related to its December 31, 2017, balance sheet. Accounts receivable .................................................... $22,000...
-
What is the risk on different financial assets and what is affecting their risk? How many different bonds and stocks exist in our financial markets? What do you think that are the most important...
-
Amanda Autry and Carley Wilson are partners in A & W Gift Shop, which employs the individuals listed below. Paychecks are distributed every Friday to all employees. Based on the information given,...
-
With reference to Problem 38, concerning opening width in the A Level, determine a bolting plan that would allow for 27.4 m wide rooms. (a) Specify, bolt length, safety factor, diameter, steel grade,...
-
A room and pillar coal mine is contemplated at a depth of 533 m in strata striking due north and dipping 18? east. Entries are driven on strike, crosscuts up, and down dip. Mining height is 4.6 m...
-
A _______________ _______________ argument is used by a court when it is concerned that a decision for a sympathetic plaintiff may lead to innumerable individuals in similar situations seeking...
-
Assume that a large managed care company has a low return on equity (ROE). How could Du Pont analysis be used to identify possible actions to help boost ROE?
-
How much of the return variance on a foreign stock investment is likely to come from variation in the foreign stock market and how much from the variation in the exchange rate? What are the...
-
Fargo Memorial Hospital has annual net patient service revenues of $14,400,000. It has two major third-party payers, plus some of its patients are self-payers. The hospitals patient accounts manager...
-
Milwaukee Surgical Supplies, Inc., has gross sales for the year of $1,200,000. The collections department estimates that 30 percent of the customers pay on the tenth day, 40 percent pay on the...
-
The country of Pepper-land exports steel to the Land of Submarines. Information for the quantity demanded (Qd) and quantity supplied (Qs) in each country, in a world without trade, are given in Table...
-
Let $a>0$ and consider the risk measure $ho(X)=\mathrm{E}[-X]+a \sigma(X)$. Is this a coherent risk measure? If not, which axioms are violated?
-
Write out the formula for the total costs of carrying and ordering inventory, and then use the formula to derive the EOQ model. Andria Mullins, financial manager of Webster Electronics, has been...
-
The engine of a large ship does 2.00 10 8 J of work with an efficiency of 5.00%. (a) How much heat transfer occurs to the environment? (b) How many barrels of fuel are consumed, if each barrel...
-
Without bracing, a machinist can exert only about 100 lbf on a wrench or tool handle. The lever shown in the figure has t = ½ in and w = 2 in. We wish to specify the fillet-weld size to secure...
-
Find Io in the circuit shown using nodal analysis. 10k 2I 2mA 101 Iz Io
-
Find in the network shown using nodal analysis. 10 k 4000 I
-
The new bookkeeper at Karlin Construction Company was asked to write off two accounts totaling $1,680 that had been determined to be uncollectible. Accordingly, he debite- Accounts Receivable for...
-
Ammonium carbonate, (NH 4 ) 2 CO 3 dissolves in water releasing the ammonium ion, NH 4 + , and carbonate ion, CO 3 2- into solution. Both of these ions hydrolyze (react with water). The chemical...
-
a) The density of Acetic acid is 1.05 g/mL and the density ofwater is 1.0 g/mL. The molar mass of Acetic Acid is 60.052 g/mol.What is the molarity of CH3COOH in water of a 5% acidity vinegarsolution?...

Study smarter with the SolutionInn App


