An electrospray/transmission quadrupole mass spectrum of the -chain of hemoglobin from acidic solution exhibits nine peaks corresponding
Question:
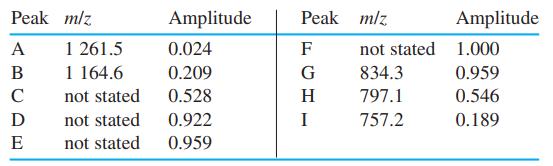
An electrospray/transmission quadrupole mass spectrum of the α-chain of hemoglobin from acidic solution exhibits nine peaks corresponding to Mn+n. Find the charge, n, for peaks A-I. Calculate the molecular mass of the neutral protein, M, from peaks A, B, G, H, and I, and find the mean value.
Transcribed Image Text:
Peak m/z Amplitude Peak mlz Amplitude 1 261.5 1 164.6 A 0.024 F not stated 1.000 В 0.209 G 834.3 0.959 C not stated 0.528 H 797.1 0.546 D not stated 0.922 I 757.2 0.189 E not stated 0.959
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The analysis follows the same steps as Table 213 The work is set out in t...View the full answer

Answered By

Umber Talat
I am providing full time mentoring and tutoring services in Business Finance, Contemporary issue in Global Economy, Quantitative Techniques, Principles of Marketing, strategic marketing, International Marketing, Organizational Behavior (OB), Consumer Behavior, Sales Force Management, Strategic Brand Management, Services Marketing, Integrated Marketing Communication (IMC), Principles of Management, General Management, Strategic Management, Small and Medium Enterprise Management, Innovation Management, Change Management, Knowledge Management, Strategic Planning, Operations Management, Supply Chain Management, Logistics Management, Inventory management, Total Quality Management (TQM), Productions Management, Project Management, Production Planning, Human Resource Management (HRM), Human Resource Development, Strategic HRM, Organizational Planning, Performance and Compensation Management, Recruitment and Selection, Organizational Development, Global Issues in Human Resource Management, Retail Marketing, Entrepreneurship, Entrepreneurial Marketing, International Business, Research Methods in Business, Business Communication, Business Ethics.
4.70+
158+ Reviews
236+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
The molecular ion region in the mass spectrum of a large molecule, such as a protein, consists of a cluster of peaks differing by 1 Da. This pattern occurs because a molecule with many atoms has a...
-
Cocaine metabolism in rats can be studied by injecting the drug and periodically withdrawing blood to measure levels of metabolites by HPLC-mass spectrometry. For quantitative analysis, isotopically...
-
Carbon has two stable isotopes, 126C and 136C, and fluorine has only one stable isotope, 199F. How many peaks would you observe in the mass spectrum of the positive ion of CF+4? Assume that the ion...
-
What approaches to recruitment can an employer adopt in order to create and project a positive public image?
-
Buffet Appliance Mart began operations on May 1. It uses a perpetual inventory system. During May, the company had the following purchases and sales for its Model 25 Sureshot camera. Instructions (a)...
-
Consider the circuit shown in Figure P5.56. (a) Determine \(R_{T H}, V_{T H}, I_{B Q}\), \(I_{C Q}\), and \(V_{E C Q}\) for \(\beta=90\). (b) Determine the percent change in \(I_{C Q}\) and \(V_{E C...
-
Assume the unadjusted and adjusted trial balances for Milky Way Theater, Inc., at November 30, 2010, show the following data: Requirement 1. Journalize the adjusting entries that account for the...
-
The 2011 Annual Report of Tootsie Roll Industries contains the following Information. Instructions Compute the following ratios for Tootsie Roll for 2011. (a) Asset turnover. (b) Return on assets....
-
Question 1 (30 points) A 3D infinite quantum well is a very simple model for an atom. Suppose that two cubic 3D infinite quantum wells, with cube dimension L, are joined to form one parallelepiped...
-
Cheapo Electrons is an electricity retailer. The table below shows the load that it forecast its consumers would use over a 6-h period. Cheapo Electrons purchased in the forward market and the power...
-
(a) To detect the drug ibuprofen by liquid chromatography/ mass spectrometry, would you choose the positive or negative ion mode for the spectrometer? Would you choose acidic or neutral...
-
Define the unit dalton. From this definition, compute the mass of 1 Da in grams. The mean of 60 measurements of the mass of individual E. coli cells vaporized by MALDI and measured with a quadrupole...
-
Solve each problem. Future Value Find the future value and interest earned if $56,780 is invested at 2.8% compounded (a) quarterly for 23 quarters (b) continuously for 15 yr.
-
How can good boilerplate terms and conditions of contract still fail to protect an organization against some risk?
-
How does the analysis from the supplier position and supplier preferencing models affect how a purchasing manager plans to do business with suppliers?
-
Would a centralized supply management function assign purchasing decisions to individual managers at each of an organization's locations?
-
List differences between the corruption analysis pattern described here and the traditional methods.
-
Show how to do qualitative measurements: Reuse, complexity, and coupling between classes for trust SAP.
-
Verify your answer to Exercise 34 by calculating the work for each force function. Data from in Exercise 34 The graphs show the force F i (in pounds) required to move an object 9 feet along the...
-
Refer to the data in QS 10-1. Based on financial considerations alone, should Helix accept this order at the special price? Explain.
-
Air pollution in the Mexico City metropolitan area is among the worst in the world. The concentration of ozone in Mexico City has been measured at 441 ppb (0.441 ppm).Mexico City sits at an altitude...
-
From the data in Table 18.1, calculate the partial pressures of carbon dioxide and argon when the total atmospheric pressure is 1.05 bar.
-
The average concentration of carbon monoxide in air in an Ohio city in 2006 was 3.5 ppm. Calculate the number of CO molecules in 1.0 L of this air at a pressure of 759 torr and a temperature of 22oC.
-
A machine costing $212,800 with a four-year life and an estimated $18,000 salvage value is installed in Luther Company's factory on January 1. The factory manager estimates the machine will produce...
-
Explore the application of advanced numerical methods, such as the finite element method (FEM) and the distinct element method (DEM), in simulating the mechanical behavior of granular soils and rock...
-
Can you elaborate on the principles of advanced constitutive modeling approaches, such as the use of critical state soil mechanics and advanced soil-structure interaction theories, in predicting the...

Study smarter with the SolutionInn App


