An equimolar mixture of ethane, propane, n-butane, and n-pentane is subjected to a flash vaporization at 150?F
Question:
An equimolar mixture of ethane, propane, n-butane, and n-pentane is subjected to a flash vaporization at 150?F and 205 psia. What are the expected amounts and compositions of the liquid and vapor products? Is it possible to recover 70% of the ethane in the vapor by a single-stage flash at other conditions without losing more than 5% of nC4 to the vapor? Obtain K-values fromFigure.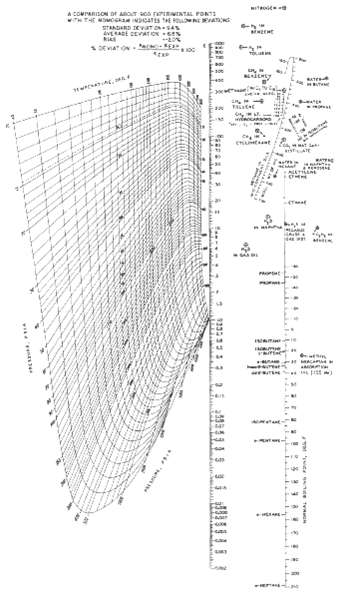
Transcribed Image Text:
A COMARISON Dr Aor acs rIENINTAL PORT1 STAADRD IEVar SAS TALate ALLTILINE
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Take as a basis a feed of 100 lbmolh From Fig at 150 o F and 205 psia the Kvalues are as given in the table below Then solve the RachfordRice equation ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
An equimolar mixture of carbon dioxide and water vapor at 1 atm and 60C enter a dehumidifying section where the entire water vapor is condensed and removed from the mixture, and the carbon dioxide...
-
An overhanging beam ABC is subjected to a couple MA at the free end (see figure). The lengths of the overhang and the main span are a and L, respectively.
-
Acme Company's production budget for August is 19,500 units and includes the following component unit costs: direct materials, $8.0; direct labor. $12.0; variable overhead. $6.0. Budgeted fixed...
-
Use a calculator to evaluate an ordinary annuity formula \[A=m\left[\frac{\left(1+\frac{r}{n}ight)^{n t}-1}{\frac{r}{n}}ight]\] for \(m, r\), and \(t\) (respectively) given in Problems 7-22. Assume...
-
Wilton Corporation reported the following information in its general ledger at December 31: All sales were on account. At the end of the year, uncollectible accounts were estimated to total $2,000...
-
Smith-Kline Company maintains inventory records at selling prices as well as at cost. For 2018, the records indicate the following data: Required: Assuming the price level increased from 1.00 at...
-
Use a dot plot to display the data set. Describe any patterns. Use the data set, which represents the pollution indices for 24 U.S. cities. 22 41 46 50 38 57 65 49 41 23 38 65 28 36 63 54 33 28 53 32...
-
A local pizza shop has hired a consultant to help it compete with national chains in the area. Because these national chains handle most business, the local shop operates as a price taker. Using...
-
1. Identifies the barriers to communication experienced in this scenario as well as their impact. 2. What are the communications barriers that Barry faces? 3. What solutions might Barry consider to...
-
Huffman codes compress text by assigning the characters that occur at the highest frequency the shortest possible codes. In this encoding scheme, no code can be a prefix of another. For example, if...
-
One hundred kilomoles of a feed composed of 25 mol% n-butane, 40 mol% n-pentane, and 35 mol% n-hexane are flashed at steady-state conditions. If 80% of the hexane is to be recovered in the liquid at...
-
The system shown in Figure is used to cool the reactor effluent and separate the light gases from the heavier hydrocarbons. K-values for the components at 500 psia and 100?F are (a) Calculate the...
-
The Walt Disney Company has four profitable business segments, described as follows: Media Networks: The ABC television and radio network, Disney channel, ESPN, A&E, E!, and Disney.com Parks and...
-
Your firm has a client, Marvin, who was injured when he ran into a disabled truck on the highway. The accident occurred shortly after sundown. Marvin said he did not see the truck until shortly...
-
Suppose Jack Major, one of your firms clients, owns a Rottweiler dog by the name of Misty, who recently bit a door-to-door salesman who came to their home. Jack is concerned that he will be sued and...
-
Your firm has a client, Ken, who purchased an Arabian stallion from Ellie Arabians. His intent was to use the stallion to enhance his breeding program, but in the first year that he used the...
-
How are awards for attorneys fees determined?
-
To how many peremptory challenges is a party entitled?
-
Describe briefly the benefits of and problems with compulsory retirement.
-
Find the velocity, acceleration, and speed of a particle with the given position function. r(t) = (t 2 , sin t - t cos t, cos t + t sin t), t > 0
-
Discuss why hedge funds in emerging markets gaining popularity and the factors that can best explain their performance.
-
A feed at 100F and 480 psia is to be separated by two ordinary distillation columns into the indicated products. Two distillation sequences (see 1.7.3) are to be examined. In the first, CH 4 is the...
-
Toluene is to be separated from biphenyl by ordinary distillation. The specifications for the separation are as follows: Temperature = 264 F; pressure = 37.1 psia for the feed; reflux ratio = 1.3...
-
A depropanizer distillation column is designed to operate at a feed stage pressure of 315 psia for separating a feed into distillate and bottoms with the following flow rates: The feed is 66 mol%...
-
You have $36,000 to Invest in Sophie Shoes, a stock selling for $60 a share. The initial margin requirement is 65 percent. calculate your rates of return if the stock rises to $90 a share and if it...
-
New Flyer Industries has decided to expand its production of hybrid transit buses. The firm expects incremental cash flows of? $40 million per year for the next 10 years. The upfront cost of the...
-
Calculate the mean, mode, median, standard deviation, and range for the Graduation Rate, Salary After Completing Program, and Annual Cost. our response to each of these should include only the mean,...

Study smarter with the SolutionInn App


