Analysis of a capillary flow meter (see Fig. 2B.8). Determine the rate of flow (in lb/hr) through
Question:
Analysis of a capillary flow meter (see Fig. 2B.8). Determine the rate of flow (in lb/hr) through the capillary flow meter shown in the figure. The fluid flowing in the inclined tube is water at 20oC, and the manometer fluid is carbon tetrachloride (CC14) with density 1.594 g/cm3. The capillary diameter is 0.010 in. Note: Measurements of H and L are sufficient to calculate the flow rate; ? need not be measured. Why?
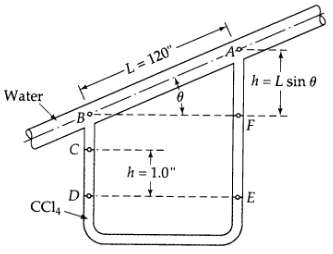
Water -Bo Co -L=120". Do CC14- 17 h = 1.0" Aº h= L sin 0 IF E
Step by Step Answer:
Designate the water by fluid I and the carbon tetrachlorid...View the full answer



Related Video
Density helps us predict whether something will float. Density is super important to consider when building things like ships and submarines. The experiment in the video examines the water density by comparing a glass of water containing sugar with a simple glass of water
Students also viewed these Chemical Engineering questions
-
Determine the rate of flow in the channel of Prob. 14.83, knowing that b = 3 m, d1 = 1.25 m, and d2 = 1.5 m. Problem 14.83: The depth of water flowing in a rectangular channel of width b at a speed...
-
Can performance on some jobs simply not be measured why or why not?
-
A Venturi tube may be used as a fluid flow meter (see Fig. 14.20). If the difference in pressure is P1 P2 = 21.0 kPa, find the fluid flow rate in cubic meters per second, given that the radius of the...
-
Samples of size n = 5 are collected from a process every half hour. After 50 samples have been collected, we calculate. x = 20.0 and s 1.5. Assume that both charts exhibit control and that the...
-
Consider four advertising campaigns where for each one it is cancelled before launch with probability 0.10, it is launched but canceled early with probability 0.18, it is launched and runs its...
-
Let S = {v1, v2, ( v"} be an ordered basis for an n-dimensional vector space V. Show that is an ordered basis for Rn. V2 {[v]s[v]s[v.J
-
Consider the gasoline mileage performance data in Table B.3. a. Use the all-possible-regressions approach to find an appropriate regression model. b. Use stepwise regression to specify a subset...
-
This problem continues the Canyon Canoe Company situation from Chapter 14. The company wants to invest some of its excess cash in trading securities and is considering two investments, The Paddle...
-
Larner Corporation is a diversified manufacturer of industrial goods. The company's activity-based costing system contains the following six activity cost pools and activity rates: Activity Cost Pool...
-
In a Q-system, the daily demand is 850 units with a standard deviation of 12 units. The Cycle service level is 97.5%. What is the reorder point?
-
Annular flow with inner cylinder moving axially (see Fig. 2B.7), a cylindrical rod of diameter KR moves axially with velocity v0 along the axis of a cylindrical cavity of radius R as seen in the...
-
Low-density phenomena in compressible tube flow2,3 (Fig. 2B.9). As the pressure is decreased in the system studied in Example 2.3-2, deviations from Eqs. 2.3-28 and 2.3-29 arise. The gas behaves as...
-
Bev and Ken Hair have been married for 3 years. They live at 3567 River Street, Springfield, MO 63126. Ken is a full-time student at Southwest Missouri State University (SMSU) and Bev works as an...
-
What are the steps in the design process?
-
What is Agile, and how is it translated into the Scrum framework?
-
One of the most important contributions of criminology to the study of fraud is: 1. The M.I.C.E. concept 2. The fraud triangle 3. The relationship of punishment to remediation 4. None of the above
-
What was the name of Enrons fair value accounting model?
-
Can the graphic highlight both the employee lacking the approval and the number of hours for each incident?
-
Apply a 3PL model (including guessing parameters) to the Law School Admission Test (LSAT) data in the R program ltm. The 2PL model may be fitted with the code ~ (F[i] ~ dnorm (0,1) dbern (pi [i,m])...
-
Determine the reactions in supports A and D and connections B and C. Sketch its shear and moment diagram and determine the magnitude ankoration of the maximum shear and moment for every member. 18 3...
-
A balloon filled with H 2 (g) at and 1.00 atm has a volume of 2.24 L. What is the final gas volume if 0.10 mol He(g) is added to the balloon and the temperature is then raised to 100 C while the...
-
How is the equilibrium real interest rate determined? How is the equilibrium nominal interest rate determined?
-
Draw a graph of the market for loanable funds. Show the effect on the equilibrium real interest rate and quantity of funds loaned and borrowed of each of the following events: a. Consumers decide to...
-
During the late 1990s, U.S. asset markets were growing rapidly, making the United States attractive to foreign savers. At the same time, strong economic growth made firms optimistic about the future,...
-
One of the two alternative long term debt or common stock will move Central Furniture Company to a more optimum capital structure. 1.what criteria are used to judge optimum capital structure?
-
Anna has a balance of $5,000 in her VISA account which grows at a nominal interest rate of 19.99% compounded daily. To pay off this balance, she makes monthly payments to VISA, with her first payment...
-
Joe works at a major pharmaceutical research and development company and has been tasked with writing his organization's data retention policy. As part of its legal requirements, the organization...

Study smarter with the SolutionInn App


