Aromatic compounds such as benzene react with alkyl chlorides in the presence of A1C1 3 catalyst to
Question:
Aromatic compounds such as benzene react with alkyl chlorides in the presence of A1C13 catalyst to yield alkylbenzenes. The reaction occurs through a carbocation intermediate, formed by reaction of the alkyl chloride with A1C13 (R-C1 + AlCl3 ? R+ + A1C14-). How can you explain the observation that reaction of benzene with 1-chioropropane yields isopropylbenzene as the major product?
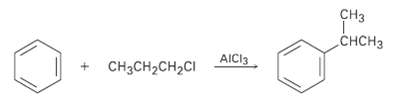
Transcribed Image Text:
сз CHCH3 AICI3 CнзCH-CH2CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
Reaction of 1chloropropane with the Lewis acid AlCl3 forms a ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Alkyl halides add to alkenes in the presence of AlCl3; yields are the highest when tertiary halides are used. Predict the outcome of the reaction of tert-pentyl chloride (1-chloro-2,...
-
In the presence of the enzyme aconitase, the double bond of aconitic acid undergoes hydration. The reaction is reversible, and the following equilibrium is established:
-
In the presence of hidden action problems, under what conditions will a deferred compensation contract both minimize taxes and provide desirable work incentives for employees?
-
Conduct some additional research to learn more about Fabletics. How is Fabletics meeting customer needs through its value delivery network? What controversy surrounds the company? What type of...
-
Identify a business or sports leader who you think is highly effective. Present your observations to the class.
-
Little Billy walks due east to school, but must cross from the south side to the north side of the street. Because he is a very careful child, he crosses quickly at the first possible opportunity. a....
-
Suggest and justify the kind of commission structure that you would put into place.
-
Develop a trend line for the demand for fertilizer in Problem 5-15, using any computer software.
-
A block is given a brief push to the right then allowed to move on a horizontal surface. The two red vertical lines in the diagram are just "markers" and do not stop the block. KL-D Vo 2 Magnitude of...
-
Are you optimistic about our ability to develop a global ethical consensus across our national and religious differences? Why or why not?
-
Epi-Aristolochene, a hydrocarbon found in both pepper and tobacco, is biosynthesized by the following pathway. Add curved arrows to show the mechanism of each step. Which steps involve alkene...
-
Alkenes can be converted into alcohols by acid-catalyzed addition of water. Assuming that Markovnikov?s rule is valid, predict the major alcohol product from each of the following alkenes. H CH3CH-CH...
-
If property is sold at a loss to a related taxpayer, under what circumstances can at least partial benefit be derived from the disallowed loss?
-
The following income statement was drawn from the records of Franklin Company, a merchandising firm: FRANKLIN COMPANY Income Statement For the Year Ended December 31, Year 1 $1,092,000 Sales revenue...
-
The December 3 1 , 2 0 2 4 , unadjusted account balances for Demon Deacons Corporation are presented below. Accounts Balance Cash $ 9 , 9 0 0 Accounts Receivable 1 4 , 9 0 0 Prepaid Rent 7 , 0 8 0...
-
Lamonda Corporation uses a job order cost system. On April 1, the accounts had balances as shown in the T- accounts below: The following transactions occurred during April: a. Purchased materials on...
-
A string of length 0.650 m and a linear density of 2.02 x 10-3 kg/m is vibrating in the fundamental harmonic mode even as the tension is increased over time. How many oscillations does the string go...
-
Sweeten Company had no jobs in progress at the beginning of March and no beginning inventories. The company has two manufacturing departments-Molding and Fabrication. It started, completed, and sold...
-
Does the asthenosphere have the same composition throughout? What is uniform throughout the asthenosphere?
-
The Cholesterol Level data sets give cholesterol levels of heart attack patients. Cholesterol measures are taken 2, 4, and 14 days aft er a patient has suffered a heart attack. Is there a significant...
-
What happens to a system in dynamic equilibrium when it is disturbed in some way?
-
Draw the more stable chair conformation for each of the following compounds: CI Cl Cl
-
Deterrtine whether each of the following compounds can in principle be isolated in optically active form under ordinary conditions. (a) 1.1 -dimethyl cyclohexane (b) cis- l -ethyl-3-methylcyclohexane
-
Draw a structure for the more stable conformation on of tmns-1,2-dimetrylcyclobutane.
-
Discuss how data deduplication works in backup systems and its impact on storage efficiency. What are some potential drawbacks of deduplication, and how might they be mitigated ?
-
Describe the importance of application consistency in backup operations. How do modern backup systems ensure consistency, and what are the risks of a quiescence failure ?
-
Find an equation in Cartesian coordinates for the equation given in cylindrical coordinates: (a) r = 2 cos 0, (b) r2 +z2 = 5. Sketch each surface in Cartesian coordinates.

Study smarter with the SolutionInn App


