As a general rule, equatorial alcohols are esterified more readily than axial alcohols. What product would you
Question:
As a general rule, equatorial alcohols are esterified more readily than axial alcohols. What product would you expect to obtain from reaction of the following two compounds with 1 equivalent of aceticanhydride?
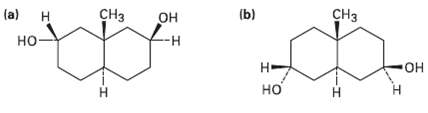
Transcribed Image Text:
СHз (b) (a) CHз он -н Н но Н- -Он но Н Н н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
a H HO b H OH H CH3 H CH3 OH H H OH 1 eq...View the full answer

Answered By

Vivek Sheoran
I am clearing the doubts in minds of students related to math and chemistry from more than an year,
My students are always getting curious about science after being taught by me.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What product would you expect to obtain from the base-catalyzed Michael reaction of (a) 1, 3-diphenylprop-2-en-1-one (Section 19.5A) and acetophenone (b) 1, 3-diphenylprop-2-en-1-one and...
-
What products would you expect to obtain from reaction of 1-methylcyclo-hexanol with the following reagents? (a) HBr (b) NaH (c) H2SO4 (d) Na2Cr2O7
-
What product(s) would you expect to obtain from reaction of 1, 3-cyclo-hexadiene with each of the following? (a) 1 mol Br2 in CH2C12 (b) O3 followed by Zn (c) 1 mol HCI in ether (d) 1 mol DCl in...
-
You are the newly appointed financial manager of The Shoebox (Pty) Ltd, which operates a chain of 20 large retail shoe stores in and around Port Elizabeth, selling on a cash-only basis. The stores...
-
1. Many speculate that Schmidt decided to release his role as CEO to Larry Page, the co-founder, to capitalize on what was previously a core competency - innovation. How would the change of CEO...
-
Design an emitter-stabilized network at ICQ = ICsat and VCEQ = 1/2 VCC = Use VCC = 20 V, ICsat = 10 mA, = 120, and RC = 4RE. Use standard values.
-
A steel manufacturer extrudes scrap for manufacturing blades. Specifications require that the thickness of this scrap has \(\mu=0.020 \mathrm{~mm}\) and \(\sigma=0.005 \mathrm{~mm}\). (a) Use the...
-
In what ways was Anderson a transformational leader in order to execute his strategy for Best Buy, how did he combine transformational with transactional leadership? In what ways did Anderson exhibit...
-
The assets and liabilities of Thompson Computer Services at March 31, the end of the current year, and its revenue and expenses for the year follow. The common stock was $118,300 and the retained...
-
Consider the deterministic world below (part (a)). Allowable moves are shown by arrows, and the numbers indicate the reward for performing each action. If there is no number, the reward is zero....
-
Draw the most stable chair conformation of menthol, and label each substituent as axial orequatorial. Menthol (from peppermint oil) - CH
-
Propose a mechanistic pathway for the biosynthesis of isoborneol. A carbocation rearrangement is needed at one point in thescheme. CH CH HO Isoborneol
-
Advocates of retrospective methodology claim there are distinguishing characteristics that increase its value over past lessons learned methods. What are they? How does each characteristic enhance...
-
Your favorite place to visit is a local microbrew. The beer here is so good that the owner of the microbrew has earned the nickname Dr. Lupulin. While engaging in small talk while enjoying your 3 rd...
-
The table gives the total emissions of lead into the atmosphere for selected years. Total Lead Emissions into the Atmosphere Year Lead, f (million tons) 1970 220.9 1975 159.7 1980 74.2 1985 22.9 1990...
-
The graph for the given constraints is shown below. Determine the corner points of the feasible region, find the value of the objective function z = 30x +25y for each of the corner points....
-
I need some help to understand how this questions could work? Setup This assignment uses a technique called "Unit Testing," and relies on a number of dependencies, which must be installed on your...
-
If Raymond has an investment offer to invest a lump sum of money now and then receive $3,500 every 6 months. What is such investment has to be made now if interest is 8% compounded quarterly and the...
-
Let \(\left(B_{t}, \mathscr{F}_{t} ight)_{t \geqslant 0}\) be a \(\mathrm{BM}^{1}\) such that \(\mathscr{F}_{0}\) contains all measurable null sets and let \(\tau\) be a stopping time. Show that...
-
Presented below are income statements prepared on a LIFO and FIFO basis for Kenseth Company, which started operations on January 1, 2024. The company presently uses the LIFO method of pricing its...
-
100 kg of steam is available at 2 MPa and 800 C. a. Determine the maximum amount of shaft work that can be obtained from this steam in a non-flow process if the ambient conditions are 25C and 1 bar....
-
Androstenedione is a steroid touted for its muscle-building ability through metabolic conversion to testosterone (page 456). What reaction must occur in the body to convert androstenedione to...
-
From the data in Table 15.2, what can you say in general about the ratio of saturated to unsaturated acids in fats and oils?
-
Design a synthesis for the amphoteric detergent shown on the preceding page, using an SN2 displacement with CH3(CH2)14CH2N(CH3)2 as the nucleophile and an appropriate halide.
-
Athens Gas Station has figured out the weekly demand distribution for their gas sales. Each gallon of gas sold at the pump results in a profit of 10 cents/gallon and any lost sales results in a cost...
-
1. The maximum pressure an eardrum can withstand without rupturing is Pmax = 3.0102 Pa. Assume a sound is made at that pressure. If the density of air is, p=1.20 kg/m, and the speed of sound is 340...
-
52. A wave is sent down a string with a linear mass density of 1.600 x 10-4 kg/m. The wavefunction that describes this wave is: y(x,t) = (0.021 m)*sin{(2.00 rad/m)*x + (30.0 rad/s)*t} What is the...

Study smarter with the SolutionInn App


