As written, the following syntheses have flaws. What is wrong witheach? CH o (b) CI (a) .CH
Question:
As written, the following syntheses have flaws. What is wrong witheach?
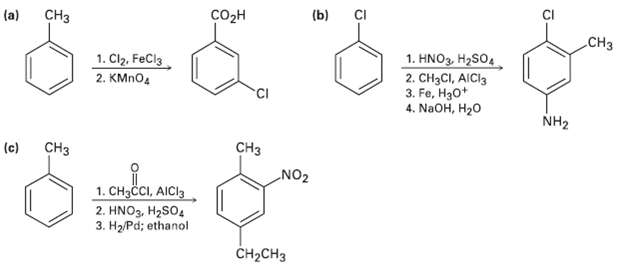
Transcribed Image Text:
CHз сoон (b) CI (a) .CHз 1. Cl2, FeCl3 1. HNO3, H2SO4 2. CH3CI, AICI3 3. Fe, H30* 4. NaOH, H20 2. KMNO4 NH2 (c) CH3 CHз NO2 1. CH3CI, AICI3 2. HNO3, H2SO4 3. H2/Pd; ethanol Cн-CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a Chlorination of toluene occurs at the ortho and Para positions To sy...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
What is wrong with these synthesesexplain. 1) NaNH2, NH, (1) C=CCH3 a) CH3C=CH 2) -Br CH;CH,-NH, I b) CH;CH,I + NH3 CI + Br OCH, CH, + CH,O d) . .. Br + HBr e) CH3 CH3 H,SO, f)
-
Syntheses of each of the following compounds have been reported in the chemical literature. Using the indicated starting material and any necessary organic or inorganic reagents, describe short...
-
What counts as cruelty to animals? Whats wrong with cruelly to animals?
-
A taxpayer has qualified business income of $50,000 from partnership A and a qualified business loss of $30,000 from partnership B. Wages allocable to the taxpayers interest in partnership A were...
-
Your client works for a defense contractor and was assigned to work on a military base in Australia. As a condition of his employment, he was required to live in housing that was provided to military...
-
A survey of 1,085 adults asked, "Do you enjoy shopping for clothing for yourself?" The results (data extracted from "Split Decision on Clothes Shopping," USA Today, January 28, 2011, p. IB) indicated...
-
How does the auditor establish the reasonableness of the allowance for uncollectible accounts?
-
Jones Products manufactures and sells to wholesalers approximately 400,000 packages per year of under-water markers at $ 6 per package. Annual costs for the production and sale of this quantity are...
-
Jay Oullette, CEO of Bumper to Bumper Incorporated, anticipates that his company's year-end balance sheet will show current assets of $12,774 and current liabilities of $7,570. Oullette has asked...
-
Molly, whose tax rate is 37%, sells an apartment complex for $4.5 million with 10% of the price allocated to land. The apartment complex was purchased in 1993. She has no other sales or exchanges...
-
Starting with either benzene or toluene, how would you synthesize the following substances? Assume that ortho and Para isomers can be separated. (a) 2-Brorno-4-nitrotoluene (b) 1, 3,...
-
How would you synthesize the following substances starting frombenzene? ( C-CH2 .CH- (a) (c) H
-
Determine the critical values of \(P_{c r}\) for which buckling will occur in the beam given in example 3.11. Data From Example 3.11: Consider a cantilever beam in figure 3.28 of length \(L=1...
-
Marc, a single taxpayer, earns $ 6 0 , 0 0 0 in taxable income and $ 5 , 0 0 0 in interest from an investment in city of Birmingham bonds. Using the U . S . tax rate schedule for 2 0 2 3 , how much...
-
The Bradford Company issued 10% bonds, dated January 1, with a face amount of $93 million on January 1, 2024. The bonds mature on December 31, 2033 (10 years). For bonds of similar risk and maturity,...
-
The value of identifying purpose as it pertains to teamwork is a way to what ? Povide meaning and significance to the work delineate personal goals from organizational goals require team members to...
-
NFLX has a beta of 1.05 and is currently selling for $79.50. If the T-bill is currently yielding 5.2% and the expected market return is 11%, does the NFLX required return based on CAPM and the...
-
For portfolios having a mean return of 25%, the minimum standard deviation of return is 29.53%. The weight on the UK index in this optimal portfolio is -110%, and is the only negative weight. Explain...
-
Describe and compare the key features of each of the following types of individual retirement arrangements. a. Traditional deductible IRA b. Roth IRA c. Nondeductible IRA d. SIMPLE IRA
-
Some people argue that the internal control requirements of the Sarbanes-Oxley Act (SOX) put U.S. companies at a competitive disadvantage to companies outside the United States. Discuss the...
-
Repeat the calculation of problem 5.22 with the vapor being recycled to the compressor Problem 5.22 Isobutane is to be liquefied to make liquid petroleum gas (LPG). The butane is available at 25 C...
-
Show the products of these reactions of 1-propylcyclopentene with thesereagents: a) Br, CCl, b) Br2, H2O d) HBr f) [OsO,], t-BuOOH c) 1) BH3, THF; 2) H,O,, NaOH e) H2O, H,SO, g) 1) O; 2) (CHz)S
-
Show the alkenes that would give these products. More than one answer may be possible in somecases. Br Br Br2 a) CCI4 Ph Ph 1) Hg(O,CCH3)2, H,O b) 2) NaBH,, NAOH CI 1) , THF d) 2) H2O2, NAOH ...
-
Show all the steps in the mechanism for these reactions. Include stereochemistry where it is important.
-
What are the facts of this case? Business Financing Solutions, Inc. ("BFS") is a small lending institution that specializes in providing loans to small businesses. BFS's president, Rachel Chen, has...
-
Royal Lawncare Company produces and sells two packaged products-Weedban and Greengrow. Revenue and cost Information relating to the products follow: Selling price per unit Variable expenses per unit...
-
This is a slip and fall case. Plaintiff was injured when she slipped and fell in the defendant's fast food restaurant. Plaintiff claims the floor was wet and slippery when she fell. Defendant denies...

Study smarter with the SolutionInn App


