Assign R or S configuration to each chirality center in prostaglandin E2 (Figure) the most abundant and
Question:
Assign R or S configuration to each chirality center in prostaglandin E2 (Figure) the most abundant and biologically potent of mammalianprostaglandin
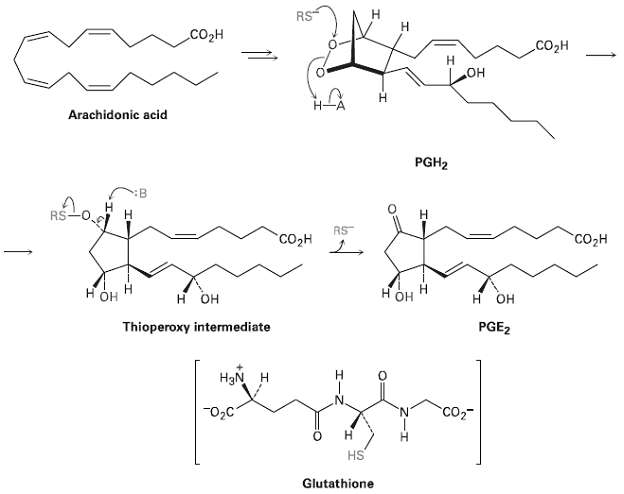
Transcribed Image Text:
н Н RS Соон .CO2H он Н Arachidonic acid PGH2 H. RS RS "СОдн "Соон нон он H OH OH PGE2 Thioperoxy intermediate HgN H "02C CO2 н HS Glutathione
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
R H I R ...View the full answer

Answered By

Michael Owens
I am a competent Software Engineer with sufficient experience in web applications development using the following programming languages:-
HTML5, CSS3, PHP, JAVASCRIPT, TYPESCRIPT AND SQL.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign R or S configuration to each chirality center in the followingmolecules: (b) NH2 (a) . "C r "O
-
Assign R or S configuration to each chirality center in the following monosaccharide?s and tell whether each is a D sugar or an L sugar: (b) (a) - (c) - - - - - - - - - - CH- CH2 CH2
-
Assign R or S configuration to each chirality center in the followingmolecules: (a) (b) ,CH (c) , . CH CHCH
-
The management accountant is preparing the master budget for her retail firm. The following information has been supplied Sales $300,000 Opening inventory $40,000 Closing inventory $60,000 Required...
-
How does an agenda help make a meeting more successful?
-
Determine vo for each network of Fig. 2.172 for the input shown. 10 V Si 2,2 k Si 5 V 1.2 k 4.7 k -10 V
-
Nitrogen gas at \(25^{\circ} \mathrm{C}\) and \(1 \mathrm{~atm}\), with \(C_{p}=7 \mathrm{cal} / \mathrm{mol}-\mathrm{K}\), is cooled to \(-100^{\circ} \mathrm{C}\) at \(1 \mathrm{~atm}\). Assuming...
-
The Akron Police Department has five detective squads available for assignment to five open crime cases. The chief of detectives, Paul Kuzdrall, wishes to assign the squads so that the total time to...
-
Determine the missing manufacturing cost data for Sunland Company as presented below. (Assume all raw materials used were direct materials.) Case A Case B Case C Direct materials used (a) $87,500...
-
The primary purpose of the reformer is to convert methane and water to carbon monoxide and hydrogen (Equation 13.1). The extent of this reaction is limited by chemical equilibrium. where Subscript...
-
Write the saponification reaction of glyceryl di-oleate monopalmitate with aqueous NaOH.
-
Studies of the conversion of mevalonate 5-phosphate to isopentenyl diphosphate have shown the following result. Which hydrogen, pro-R or pro-S, ends up cis to the methyl group, and which ends...
-
The variable manufacturing costs per unit of Nascar Motors are as follows: Required: 1. Prepare income statements for Nascar Motors in April and May 2014 under throughput costing. 2. Contrast the...
-
A Project Manager discovers that his team has neglected to complete the network diagram for the project. The network diagram is shown below but you need to fill the duration, ES/EF/LS/LF & Slack for...
-
An electroscope is a device made of a metal ball connected through a wire to two aluminum plates that are suspended inside a glass flask. A student generates electrostatic energy by rubbing a plastic...
-
The current US Dollar to Yen exchange rate is Mo 110. The CCIR US rate is 4% and the CCIR Japanese rate is 2%. Banco Santander is currently willing to offer a one-month forward contract (assuming...
-
Describe in words why minimizing the sum of the absolute deviations for a linear model is different from minimizing the sum of the squared deviations for a linear model. How might you teach this to...
-
Conflict Styles Assessment Dr. Stella Ting-Toomey & Dr. Leeva Chung, UIC Text, pp.193-194/ my.blog 9.3 Assessing Your Specific Five Conflict Styles: Instructions: Recall how you generally communicate...
-
Show that Equations 2.57 through 2.60 are correct, using a. a cash flow diagram. b. the mathematical formulas for each of the factors given. (P|Gi%,1)=0 (A|Gi%,1) = 0 (A|P 1%,1) = (F|Pi%,1) = 1+i (AF...
-
Vectors are drawn from the center of a regular n-sided polygon in the plane to the vertices of the polygon. Show that the sum of the vectors is zero.
-
A 0.148 M solution of a monoprotic acid has a percent ionization of 1.55%. Determine the acid ionization constant (K a ) for the acid.
-
Describe the 1H NMR spectrum of a. BrCH2CH2Cl b. ClCH2CH2Cl
-
Write an equation for the reaction of pyridine with a. cold sulfuric acid (H2SO4) b. cold nitric acid (HNO3)
-
Although nitration of pyridine requires a temperature of 300°C (eq. 13.2), 2,6 dimethylpyridine is readily nitrated at 100°C. Write an equation for the reaction, and explain why milder...
-
The sheet Inventory lists a grocery store's inventory for two months. Calculate the percent change for each item to two decimal places. If the formula gives an error, put "Initial Stock" in the cell....
-
A local manufacturer produces two different products on the same production line. Product A requires .5 hours of labor while product B requires .8 hours of labor. The total labor available in any one...
-
Hampton Company reports the following information for its recent calendar year. Income Statement Data Sales Expenses: Cost of goods sold Salaries expense Depreciation expense Net income Required:...

Study smarter with the SolutionInn App


