Assume that acetyl COA containing a 14C isotopic label in the carboxyl carbon atom is used as
Question:
Assume that acetyl COA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in lanosterol where the label wouldappear.
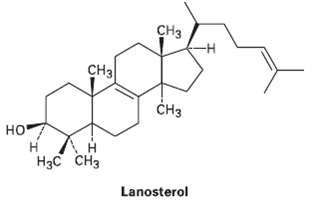
Transcribed Image Text:
CHз Cнз CHз но° н н Нзс снз Lanosterol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Squalene is converted to lanosterol by the series of steps pictured in Figure Ext CH3 CH3 p...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in ?-cadinol where...
-
Identify the carbon atoms expected to be labeled with 14C when each of the following substances is biosynthesized from acetate enriched with 14C in its methyl group: (a) (b) (c) (d) Palmitic acid CH3...
-
Label each hydrogen atom in the following compounds as primary (1°), secondary (2°), or tertiary (3°). (a) CH3 CH2 CH (CH3)2 (b) (CH3)3 CCH2CH3 (c) (CH3)2 CHCH (CH3) CH2 CH3 (d) (e) (f) ...
-
Wilburton Hospital is investigating the possibility of investing in new dialysis equipment. Two local manufacturers of this equipment are being considered as sources of the equipment. After-tax cash...
-
What must strategic leaders do to develop and sustain an effective organizational culture?
-
For the collector-feedback configuration of Fig. 4.119, determine: (a) IB. (b) IC. (c) VC. +16 V 3.6 k 470 k p- 120 0.51 k
-
Suppose that with the samples of Exercise 15.6, it is desired to establish control also over the variability of the process. Using the method of Exercise 15.5 and the values of \(\overline{\bar{x}}\)...
-
The MoMi Corporations cash flow from operations before interest and taxes was $2 million in the year just ended, and it expects that this will grow by 5% per year forever. To make this happen, the...
-
Pharoah Company entered into these transactions during May 2025, its first month of operations. 1. Stockholders invested $44,500 in the business in exchange for common stock of the company. 2....
-
Consider the blending system shown in Figure. A feedback control system is used to reduce the effect of disturbances in feed composition, x 1 , n the controlled variable, product composition, x....
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in squalene where...
-
Propose a mechanistic pathway for the biosynthesis of caryophyllene, a substance found in cloveoil. Caryophyllene H3C-
-
Complete the following chart by placing an "X" under the applicable column headings. Classify each cost as a fixed cost or a variable cost and as either a direct or indirect product cost or a period...
-
(1) Show that the following languages are not regular using Pumping Lemma. L = {ww: wl (a, b)* (2 points) L = {aba: p n+1} (with n 0,10 ) (2 points) (2) 5.2.13 (P145) (2 points) (3) Find a Context...
-
Find a possible source to validate the CHEMCAD model with suggestions on what thermodynamic to use. Variation for H2/H20 separation. Basically summarized what process take place, what thermodynamic...
-
Question 4, Sensitivity Analysis (15 marks) Kathleen Allen has up to $70,000 to divide among several investments. The alternative investments are municipal bonds (MB) with an 8.5% annual return,...
-
An android application that will get the user's guess number and will be compared to the computers' random rolled dice. At the start of the game, the user has a 20-point credit. If the guess is...
-
A flat, homogeneous plate of mass m and circular sector shape of radius R and angle 3/2 is given, as shown in the following figure. y It is requested: 1. Coordinates of center of mass, G. 2. Inertia...
-
Let \(\left(\mathcal{C}_{(\mathrm{o})}, \mathscr{B}\left(\mathcal{C}_{(\mathrm{o})} ight), \mu ight)\) be the canonical Wiener space (we assume \(d=1\) ). We will now consider the space...
-
Write each fraction as a percent. 7 50
-
Calculate [OH ] in each aqueous solution at 25 C, and classify the solution as acidic or basic. a. [H3O] = C. [H3O+] = 1.2 x 10-8 M 3.5 x 10- M b. [H3O] = 8.5 x 10-5 M
-
Complete the equation for each of the following reactions: a. b. CiSH3 CO K* + HCI
-
In 1789, the year of the French Revolution, the young chemist Nicolas Leblanc developed the first method for the inexpensive mass production of sodium carbonate, Na2CO3. His discovery ultimately made...
-
Using eq. 15.9 as a model, write equations for the preparation of an alkylbenzenesulfonate synthetic detergent, starting with 1-dodecene and benzene.
-
Factory overhead of $44,700 consists of Indirect labor of $21,900, Depreciation expense-Factory of $16,900, and Factory utilities of $5,900. a. Compute total manufacturing costs. b. Prepare a...
-
In Giannakakos et al. (2016), titled Using Video Modeling with voiceover Instruction plus feedback to train staff to implement direct teaching procedures, why do you think collecting data on the...
-
Presidio, Inc., produces one model of mountain bike. Partial information for the company follows: Required: 1. Complete Presidios cost data table. 2. Calculate Presidios contribution margin ratio and...

Study smarter with the SolutionInn App


