Assume that the following compound has the S configuration at its asymmetric carbon. Could this compound be
Question:
Assume that the following compound has the S configuration at its asymmetric carbon.
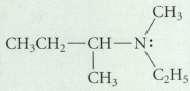
Could this compound be resolved into enantiomers?
Transcribed Image Text:
CHj CH CH2 -CHN: CH CHs
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
Each of the rapidly interconverting species in part a ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A classmate tells you that the following compound has the name 3-propylhexane. a. Is he right? If not, what error did he make and what is the correct name? b. How could you redraw the condensed...
-
The following compound has two aromatic rings. Identify which ring is expected to be more reactive toward an electrophilic aromatic substitution reaction.
-
The following compound has two isomers: ClCH==CHCl One isomer has a dipole moment of 0 D, and the other has a dipole moment of 2.95 D. Propose structures for the two isomers that are consistent with...
-
(a) Write the chemical reactions whose equilibrium constants are Kb and Ka for imidazole and imidazole hydrochloride, respectively.
-
Create a chart in which you describe the stages of Piagets theory of cognitive development. For each stage, identify a task that a child could perform.
-
(a) Find a conversion factor to convert from miles per hour to kilometers per hour. (b) In the past, a federal law mandated that highway speed limits would be 55 mi/h. Use the conversion factor of...
-
With reference to Exercise 10.9, how would the required sample size be affected if it is known that the proportion to be estimated is at least 0.75 ? Data From Exercise 10.9 10.9 What is the size of...
-
Given the following data, on a separate sheet of paper create monthly and yearly income statements for this fast-food restaurant in New York City. a. Sales for the month of June were $300,000. Sales...
-
Explain the role of policy in addressing the injustice in community health facilities.?
-
As a financial analyst at Glencolin International (GI) you have been asked to revisit your analysis of the two capital investment alternatives submitted by the production department of the firm....
-
Which of the following compounds could in principle be resolved into enantiomers at very low temperatures? Explain. (a) Propane (b) 2,2,3,3, -tetramethylbutane
-
Draw a Newman projection of the confirmation of (2S,3R,4S)-2,3,4-hexanetriol (shown above) About the C2{3 bond, with C2 nearest the observer
-
Choose the correct verb form. There (is/are) hardly even a speck of dirt left on the carpet.
-
Consider the summary output of the following two regressions where the analysis is separated for the American and National leagues. (Some of the output is omitted to save space.) American League:...
-
Practice 2 Stock market (20 points) 1. Go to the Industry Center in Yahoo Finance! 2. Choose an industry and a specific company within this industry. 3. Briefly introduce the company you chose,...
-
Florida Chocolate Inc. was first opened as a sole proprietor in 2 0 x 2 under the name Alexandria s Chocolates. The company produced 4 specialty chocolate products using an old family recipe, which...
-
n n Curves of the form x = "= cos(t) + 1cos((n = 1)t), y = n= sin(t) + sin((n-1)t) are called hypocycloids. Plot the following three curves on the same axes. Use different colors to identify...
-
A pharmaceutical company is testing a new drug. Of the 110 people that received the drug, 61 of the people's autoimmune disease went into remission. Using the old drug the autoimmune disease for 50%...
-
A 2017 Gallup Poll1 survey regarding Americans concerns about environmental issues revealed the following: Given results of surveys like the one above, it shouldnt be surprising that more and more...
-
Explain the operation of the dividends received deduction.
-
The H of hydrogenation is the heat liberated when a compound undergoes catalytic hydrogenation. Consider the H values for hydrogenation of the following three alkenes: 3-methyl -1 -butene, 2126.8 kJ...
-
The heat of formation of (E)-1,3-pentadiene is 75.8 kJ mol 1 (18.1 kcal mol 1 ), and that of 1,4-pentadiene is 106.3 kJ mol 1 (25.4 kcal mol 1 ). (a) Which alkene has the more stable arrangement of...
-
Invoking Hammonds postulate, draw the structure of the reactive intermediate that should most closely resemble the transition state of the rate-limiting step for the hydration of 1-methylcyclohexene....
-
What are the challenges and strategies for implementing TPM in highly regulated industries, such as pharmaceuticals or aerospace? How can TPM contribute to compliance and quality assurance in these...
-
In multi-site manufacturing operations, what role does TPM play in standardizing maintenance practices and ensuring consistency across different plants or facilities? Discuss the challenges and...
-
Identify and evaluate the components of a comprehensive total rewards and motivation system? Illustrate with specific examples.

Study smarter with the SolutionInn App


