At 20?C. 1,000 kg of a mixture of 50 wt% Na2S04 . 10H2O and 50 wt% Na2S04
Question:
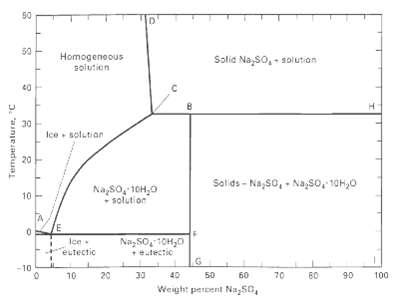
At 20?C. 1,000 kg of a mixture of 50 wt% Na2S04 . 10H2O and 50 wt% Na2S04 crystals exists. How many kilograms of water must be added to just completely dissolve the crystals if the temperature is kept at 20°C and equilibrium is maintained? UseFigure.
Transcribed Image Text:
50 50 Homogeneous solution Solia Na, so,- solution Ice solutian 20 Salids - Na,S0, - Na,So, 10H,0 Na,8o, 10H,0 solution 10 Na, SO, 10H,0 Ice eutectic +ealectio -10 60 10 20 30 40 50 60 70 90 100 Weight percent Na,50, Temperalure, "C 30
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
From Fig the solubility of Na 2 SO 4 in water at 20 o C is 15 wt ...View the full answer

Answered By

Jeff Omollo
As an educator I have had the opportunity to work with students of all ages and backgrounds. Throughout my career, I have developed a teaching style that encourages student engagement and promotes active learning. My education and tutoring skills has enabled me to empower students to become lifelong learners.
5.00+
5+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
How many kilograms of nickel must be added to 1.75 kg of copper to yield a liquids temperature of 1300(C?
-
How many kilograms of nickel must be added to 5.43 kg of copper to yield a solidus temperature of 1200(C?
-
How many grams of MgO must be added to 1 kg of NiO to produce a ceramic that has a solidus temperature of 2200 C?
-
Why is it likely to have preexisting normal faults in an orogenic belt?
-
Tarika Ltd. is a profitable small business. It has not, however, given much consideration to internal control. For example, in an attempt to keep clerical and office expenses to a minimum, the...
-
Pam Corporation acquired a 70 percent interest in Sun Corporation's outstanding voting common stock on January 1, 2016, for $980,000 cash. The stockholders' equity (book value) of Sun on this date...
-
Go to the library and find five journal articles in your area of interest in which reliability and validity data are reported. Discuss the outcome measures that are used. Identify the type of...
-
Iceland, Inc., is a fast-growing ice-cream maker. The company's new ice-cream flavor, Cherry Star, sells for $9 per pound. The standard monthly production level is 300,000 pounds, and the standard...
-
Company Y acquired The Seoul Post in February 2021 for $315 million. Suppose the yearly margin per Seoul Post reader is $1 per year (coming from advertising), and the yearly retention rate is 70%,...
-
On February 12, 2004, Nancy Trout and Delores Lake formed Kingfisher Corporation to sell fishing tackle. Pertinent information regarding Kingfisher is summarized as follows. ¢ Kingfisher's...
-
Repeat Example 4.11, except let the original solution be 20 wt% Na2S04.
-
Repeat Example 4.12, except determine the grams of activated carbon to achieve: (a) 75% adsorption of phenol. (b) 90% adsorption of phenol. (c) 98% adsorption of phenol.
-
Sewell and Reilly owned adjoining lots. They entered into a written agreement whereby each agreed to allow the other to use the south 10 feet of his lot for alley purposes for so long as the alley...
-
On 1 January 2015 the following balances, among others, stood in the books of A. Cook, a sole proprietor: (a) Business rates, 600 (Dr); (b) Packing materials, 1,400 (Dr). During the year ended 31...
-
D. Faculti started in business buying and selling law textbooks, on 1 January 2016. At the end of each of the next three years, his figures for accounts receivable, before writing-off any bad debts,...
-
Derive an expression for each of the following that is, as much as possible, in terms of measurable properties: and C V , and their partial derivatives with respect to each other. S can appear in the...
-
J. Wright, a sole trader, extracted the following trial balance from his books at the close of business on 31 March 2016: Required: Prepare the statement of profit or loss for the year ending 31...
-
On 1 January 2015 a business purchased a laser printer costing 1,800. The printer has an estimated life of four years after which it will have no residual value. It is expected that the output from...
-
A person with type AB blood has a child with a person with type O blood. What are the possible blood types of the child? a. A or B b. A, B, or O c. A, B, AB, or O d. O only
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
Explain what is meant by push and pull factors in relation to turnover. What can an organization do, if anything, to control these factors?
-
What is diafiltration? How does it differ from continuous feed-and-bleed ultrafiltration? Under what conditions is diafiltration used in conjunction with continuous feed-and-bleed ultrafiltration?
-
In Example 8.19, the % extraction of lysozyme is high (95.2%), but the % purity of the lysozyme in the total extracted proteins is low (15.1%). The Kremser plot of Figure 5.9 suggests that by...
-
What is continuous feed-and-bleed ultrafiltration? What are its limitations?
-
Please code in Python def reflect_points(line: list[tuple[int, int]], n: int) -> list[tuple[int, int]]: """ Return the given reflected vertically across the middle horizontal of an n-by-n...
-
Over the past 40 years, union membership has declined, and it continues to do so. Instead, many companies are turning to alternative dispute resolution. We know one of the best union avoidance...
-
how do you put in favourable and unfavorable in an excel spreadsheet ?

Study smarter with the SolutionInn App


