At 25C and 101 kPa, 2 mol of a gas containing 35 mol% propylene in propane is
Question:
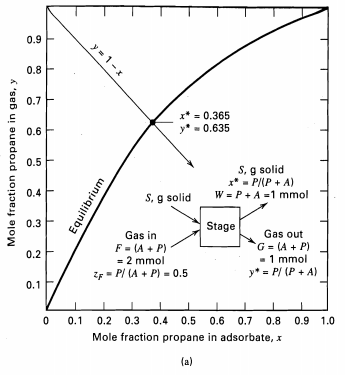
At 25°C and 101 kPa, 2 mol of a gas containing 35 mol% propylene in propane is equilibrated with 0.1 kg of silica gel adsorbent. Using the equilibrium data of Figure, calculate the moles and composition of the gas adsorbed and the equilibrium composition of the gas not adsorbed.
Transcribed Image Text:
0.9 y = 1-x 0.8 0.7 x* = 0.365 - 0.635 0.6 0.5 S, g solid x* - P/(P + A) W = P+A =1 mmol 0.4 S, g solid 0.3 Stage Gas out G = (A + P) - 1 mmol y* = P/ (P + A) Gas in F = (A + P) = 2 mmol 0.2 ZF- P/ (A + P) = 0.5 0.1 0.1 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 Mole fraction propane in adsorbate, x (a) Mole fraction propane in gas, y wnuqinb3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
Let y and x be the mole fractions of P in the vapor and ad...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
A gas containing 50 mol% propylene in propane is to be separated with silica gel having the equilibrium properties shown in Figure. The final products are to be 90 mol% propylene and 75 mol% propane....
-
A 30.0-liter cylinder of a gas containing 97.0 mole% CO and 3.0% CO2 is delivered to your plant. You sign the receipt for it, noting that the gauge on the tank reads 2000 psi. Several days later you...
-
The temperature of 3.00 mol of a gas with Cv = 6.00cal/mol K is to be raised 50.0 K. If the process is at constant volume, what are? (a) The energy transferred as heat Q, (b) The work W done by the...
-
Why do joints tend to be evenly spaced rather than clustered?
-
The following information is for Hudson Corporation. HUDSON CORPORATION Bank Reconciliation August 31 ..........................................................................$41,720 Add: Deposits...
-
Pop Corporation acquired a 90 percent interest in Son Corporation's outstanding voting common stock on January 1, 2016, for $630,000 cash. The stockholders' equity of Son on this date consisted of...
-
Data Analysis tools to compute the correlations between hand-washing efficiency scored on a scale of 1 to 5 with 1 being best (Wash), number of times hands are washed each day (Number), and number of...
-
Assume that Stanford CPAs encountered the following issues during its various audit engagements in 2014: 1. It conducted the audit of Luck, a new client this past year. Last year, Luck was audited by...
-
Consider an unknown semiconductor. At 270 K, you measure a resistance of 0.01 Ohms, while at a temperature of 300 K, you measure a resistance of 2.591 10-4 Ohms. 1) What is the gap of this material,...
-
Star Videos, Inc., produces short musical videos for sale to retail outlets. The company's balance sheet accounts as of January 1 are given below. Because the videos differ in length and in...
-
Nitrogen at 760 torr and 300?C contains 10 mol% anthraquinone (A). If this gas is cooled to 200?C, calculate the percent desublimation of A. Vapor pressure data for solid A are as follows: These data...
-
Repeat Example 4.17 for 90% evaporation of the water.
-
Since the Hawk-Eye instant replay system for tennis was introduced at the U.S. Open in 2006, men challenged 2441 referee calls, with the result that 1027 of the calls were overturned. Women...
-
(a) Identify the four factors which cause non-current assets to depreciate. (b) Which one of these factors is the most important for each of the following assets? (1) a gold mine (2) a van {3) a...
-
In your own words, explain the logic behind Formula (1). Formula 1 P(x) = (C) (N-KCn-x) NCn (1)
-
In Michigans Classic Lotto 47 Lottery, a player must choose six numbers between 1 and 47, inclusive. Six balls numbered from 1 to 47 are then randomly selected from an urn. The random variable X...
-
The following figures relate to the retail business of A. Bell for the month of July 2016. Goods which are on sale fall into two categories, X and Y. You are to calculate for each category of goods:...
-
J. Almeida is a trader who sells all of her goods at 40% above cost. Her books give the following information at 31 December 2017: You are required to: (a) Ascertain cost of goods sold. (b) Show the...
-
A cross is made between a white-eyed female fruit fly and a redeyed male. What would be the reciprocal cross? a. Female is XwXw and male is XwY. b. Female is Xw+Xw+ and male is Xw+Y. c. Female is...
-
In Problems, solve each system of equations. x + 2y + 3z = 5 y + 11z = 21 5y + 9z = 13
-
Explain the effects of the global economic crisis on employment.
-
What is the basic equation for computing the rate of mass transfer through a membrane? Explain each of the four factors in the equation and how they can be exploited to obtain high rates of mass...
-
For the commercial application of membrane separators discussed at the beginning of this chapter, calculate the permeabilities of hydrogen and methane in barrer units.
-
What kinds of materials are membranes made from? Can a membrane be porous or nonporous? What forms pores in polymer membranes?
-
The recent collapse of two major banks, Silicon Valley Bank and Signature Bank, and the subsequent federal intervention to protect uninsured depositors, has reignited debates about government...
-
Privatizing Social Security in the United States has been a topic of debate for many years, with strong opinions on both sides since its creation as part of President Franklin D. Roosevelt's New...
-
Economists measure the price level by using a basket of goods and services and calculating how the total cost of buying that basket of goods will increase over time. Economists often express the...

Study smarter with the SolutionInn App


