At what approximate positions might the following compounds show IRabsorptions? (c) (b) CH CH (a) CH3CH2CH3 CHCCH2CH3CH2
Question:
At what approximate positions might the following compounds show IRabsorptions?
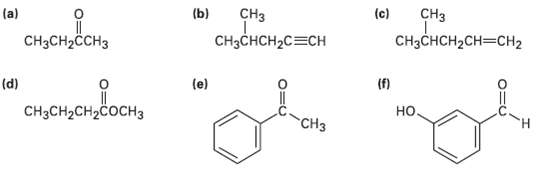
Transcribed Image Text:
(c) (b) CHз CHз (a) CH3CH2CH3 CHзCнCH2CH3CHН2 CHзснCH2C3CH (f) но. (d) (e) "онне CнзCH2CH2COCHз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
a b c d e Compound CH3CHCCH3 CH32CHCHCCH CH32CHCHCHCH CH3CHCHCOCH3 i CCH3 mol HO O Di...View the full answer

Answered By

Muhammad Ghyas Asif
It is my obligation to present efficient services to my clients by providing a work of quality, unique, competent and relevant. I hope you have confidence in me and assign me the order and i promise to follow all the instructions and keep time.
4.60+
109+ Reviews
203+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
At what approximate positions might the following compounds show IRabsorptions? c??H? " alt="At what approximate positions might the following compounds show IRabsorptions? 51524" class="fr-fic...
-
The following compounds show different rates of debromination. One reacts quite fast, and the other seems not to react at all. Explain this surprising difference in rates. Br KI, acetone Br (CH),C RS...
-
Which of the following compounds show only a single peak in their 1H NMR spectrum? a. CH3CH2OCH2CH3 b. c. CH,CH,CCI
-
Fill in the missing amounts for the following bank reconciliation: Practice Exercise3 Bank Reconciliation March 31,20 Bank Statement Balance $3,764.00 Add: Deposit in transit $4,031.00 Deduct:...
-
Why does Sutton's model apply so well to the consumer goods market? Does Sutton's model describe the structure of other markets?
-
The iodine bromide molecule, IBr, is an inter-halogen compound. Assume that the molecular orbitals of IBr are analogous to the homo nuclear diatomic molecule F2. (a) Which valence atomic orbitals of...
-
Why is the evaluation of a projects success or failure somewhat subjective?
-
Consider the following transactions that occurred in September 2012 for Aquamarines, Inc. Sep 3 Purchased inventory on terms 1/15, n/eom, $5,000. 4 Purchased inventory for cash of $1,700. 6 Returned...
-
Way Cool produces two different models of air conditioners. The company produces the mechanical systems in its components department. The mechanical systems are combined with the housing assembly in...
-
K. Nelson, project manager of Print Software, Inc., wants you to prepare a project network; compute the early, late, and slack activity times; determine the planned project duration; and identify the...
-
How would you use infrared spectroscopy to distinguish between the following pairs of constitutionalisomers? (a) CH3C=CCH3 and CHCH2C3CH () CHH CHCCH2HH2 and (c) H CH3CH2CHO and
-
Assume you are carrying out the dehydration of 1-methylcyclohexanol to yield 1-methykyclohexene. How could you use infrared spectroscopy to determine when the reaction is complete?
-
What types of changes is Chobani planning to establish itself as a company?
-
What are four elements of film theory or techniques being used in the film Citizen Kane to produce meaning with examples?
-
Explain how you felt while doing the in-class role play activity (be specific): What was challenging? What was easy? Did you work well with your partner(s)? Why/why not? How did acting out a...
-
Why does Gucci produce most of its products in Italy rather than in Southeast Asia? How does this strategy help Gucci maintain its reputation for ethics and sustainability? What does Gucci's...
-
Analyze In the space below: 1. Define the Purpose of your Report. 2. Determine Who your Primary Audience is. a) Consider the extent to which it might go to someone else. What if it does? 3. ...
-
How can you improve communication and encourage input from your employees without repercussion?
-
Can an object be accelerated without changing its kinetic energy?
-
In the simple quantity theory of money, what will lead to an increase in aggregate demand? In monetarism, what will lead to an increase in aggregate demand?
-
An isotonic solution contains 0.90% NaCl mass to volume. Calculate the percent mass to volume for isotonic solutions containing each solute at 25 C. Assume a vant Hoff factor of 1.9 for all ionic...
-
Draw the organic products you would expect to isolate from the following reactions (after hydrolysis). (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (m) (n) (o) (CH2 = CH)2 CuLi + CH3CH2CH= CHCH2Br...
-
Show how you would use Grignard syntheses to prepare the following alcohols from the indicated starting materials and any other necessary reagents. (a) octan-3-ol from hexanal, CH3 (CH2)4CHO (b)...
-
Give a systematic (IUPAC) name for each diol. (a) CH3CH(OH)(CH2)4 CH(OH)C(CH3)3 (b) HO--(CH2)8-OH (C) (d) (e) HO HO
-
What challenges arise when the HRM is uninformed concerning their legal rights and responsibilities? What is the potential impact on the organization? What should employees' expectations be...
-
Elaborate on "The Value of Learning of Applied Research Methods in Sports Management or Kinesiology" as another section of your Applied Research Methods in Sports Management. source:...
-
1. A entered into a contract with B, who is a minor. Can A later asks for the invalidation of the contract by reason of the minority of B? 10 points 2. A, the seller, and B, the buyer, agreed for the...

Study smarter with the SolutionInn App


