Azulene, a beautiful blue hydrocarbon, is an isomer of naphthalene. Is azulene aromatic? Draw a second resonance
Question:
Azulene, a beautiful blue hydrocarbon, is an isomer of naphthalene. Is azulene aromatic? Draw a second resonance form of azulene in addition to thatShown.
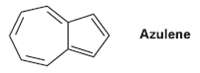
Transcribed Image Text:
Azulene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Azulene Azulene is aroma...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Naphthalene is colorless, but its isomer azulene is blue. Which compound has the lower-energy pi electronic transition? naphthalene azulene
-
Azulene is a deep-blue hydrocarbon with resonance energy of 205 kJ mol (49 kcal mol), Azulene has ten pi electrons, so it might be considered as one large aromatic ring. Its electrostatic potential...
-
Draw another resonance form for each of the substituted allylic cations shown in the preceding figure, showing how the positive charge is shared by another carbon atom. In each case, state whether...
-
Data Set 14 in Appendix B lists highway fuel consumption amounts (mi/gal) for cars categorized by size (small, midsize, large). If we use those highway fuel consumption amounts arranged into the...
-
Why is it important for ATS to recruit and train young workers instead of only hiring experienced employees from factories that are closing?
-
Propose structures for the compounds G and H whose 1H NMR spectra are shown in Figs. 9.47 and 9.48. TMS 4.2 4.0 2.0 1.5 1.0 7 6 4 2 0 (ppm) H, C3H4Br2 TMS 6.0 5.8 5.64.4 4.2 4.0 7 6 4 0 (ppm)
-
Galway Kinnell plans to apply an attributes estimation sampling plan to the shipping controls used by the Raffel & Olds Corporation, a December 31, 1999 year end printing company in New York. Based...
-
1. Why does this attempt at project partnering appear to be failing? 2. If you were Karin, what would you do to get this project back on track? 3. What action would you take to keep the project on...
-
Stock X had a return of 7.00% last year. Stocks Y and Z earned -3.00% and 11.10% respectively. The following table shows 3 portfolios: A, B, and C. What is the portfolio return under the following...
-
Recording a Business Combination Petra Corporation paid $ 8 million in cash to acquire the assets and liabilities of Salisbury Company. Petra also agreed to make an additional cash payment in the...
-
Show the relative energy levels of the seven molecular orbitals of the Cycloheptatrienyl system. Tell which of the seven orbitals are filled in the cation, radical, and anion, and account for the...
-
How many electrons does each of the four nitrogen atoms in purine contribute to the aromatic ? system? N. Purine
-
What are the codes for a, e, i, k, o, p, and u if the coding scheme is represented by this tree? 0 0 0 0 0
-
1-ABC Company had packaging costs of $170000 when 12500 packages were shipped. Packaging costs were $190000 when 19500 packages were shipped.Compute the total shipping cost when 15000 units are...
-
The Kuwait Finance House (KFH) had the following transactions during the month of July 2015: 1) A customer deposited KD 2,000 cash and KD 5,000 check drawn on another bank to his saving account. 2)...
-
The following cost data pertain to the operations of Montgomery Department Stores, Incorporated, for the month of July. Corporate legal office salaries $ 6 2 , 2 0 0 Apparel Department cost of sales...
-
Novak Corporation's statement of financial position as at December 31, 2023, showed the following amounts: Cash $140; Accounts Receivable $610; Land $1,050; Accounts Payable $430; Bonds Payable $470;...
-
john have been given cost of goods sold in the income statement for last year, but nothing has been said about purchases this year except that 1 percent was procured in august, the company buys in...
-
The first-order \(x\)-ray diffraction of monochromatic \(x\) rays from a crystal occurs at angle \(\theta_{1}\). The crystal is then compressed, causing a slight reduction in its yolume. Does...
-
From a medical tourist perspective, compare Shouldice with the traditional hospital in terms of the key factors of competition. Using Table 15-3, why would Shouldice attract patients from outside the...
-
For the reaction N 2 O 4 (g) 2 NO 2 (g), a reaction mixture at a certain temperature initially contains both N 2 O 4 and NO 2 in their standard states, which means that P N2O 4 = 1 atm and P NO 2 =...
-
Give an equation to show the reduction of Tollens reagent by maltose.
-
Does lactose mutarotate? Is it a reducing sugar? Explain. Draw the two anomeric forms of lactose.
-
Trehalose is a nonreducing disaccharide (C12H22O11) isolated from the poisonous mushroom Amanita muscaria. Treatment with an -glucosidase converts trehalose to two molecules of glucose, but no...
-
Alice is single and self-employed in 2023. Her net business profit on her Schedule C for the year is $192,800. What are her self-employment tax liability and additional Medicare tax liability for...
-
D.L. Ko and Company, a manufacturer of quality handmade walnut bowls, has had a steady growth in sales for the past 5 years. However, increased competition has led Mr. Ko, the president, to believe...
-
McCullough Hospital uses a job-order costing system to assign costs to its patients. Its direct materials include a variety of items such as pharmaceutical drugs, heart valves, artificial hips, and...

Study smarter with the SolutionInn App


