Base treatment of the following ?, ?-unsaturated carbonyl compound yields an anion by removal of H from
Question:
Base treatment of the following ?, ?-unsaturated carbonyl compound yields an anion by removal of H from the y carbon. Why is hydrogen?s on the y carbon atom acidic?
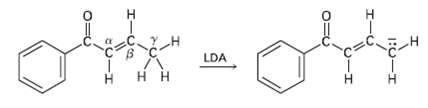
Transcribed Image Text:
LDA 1:0-I I.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
Enolization at the y position produc...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Treatment of the following alkene with a peroxyacid yields an epoxide different from that obtained by reaction with aqueous Br2 followed by base treatment. Propose structures for the two epoxides,...
-
Treatment of the following epoxide with aqueous acid produces a carbocation intermediate that reacts with water to give a diol product. Show the structure of the carbocation, and propose a mechanism...
-
An carbonyl compound can be prepared by a reaction known as a selenenylation-oxidation reaction. A selenoxide is formed as an intermediate. Propose a mechanism for the reaction. 1. LDA/THF 2. CgHSeBr...
-
On January 1, 2012, Wilmes Floral supplies borrowed $2,413 from Bower Financial Services. Wilmes Floral Supplies gave Bower a $2,500 note with a maturity date of December 31, 2013. The note specified...
-
What are some tips for business writers wishing to apologize effectively?
-
Parking fees at a zoo are $5.00 for local residents and $7.50 for all others. At the end of each day, the total number of vehicles parked that day and the gross receipts for the day are recorded, but...
-
Implement a Bayesian belief network in the programming language of your choice to represent a subject in which you are interested (for example, you might use it to diagnose medical conditions from...
-
Urdan Co. distributes suitcases to retail stores and extends credit terms of 1/10, n/30 to all of its customers. At the end of June, Urdans inventory consisted of suitcases costing $1,200. During the...
-
Find the Fg and % Diff of Fg and Fc Mass of hanging weight (kg) Time of 10 revolutions (second) Circumference (cm) Tangential velocity of the stopper(2r/t) The Fg of the hanging weights ...
-
Sanderson is a bus driver who earns a monthly gross income of $3,500. His wife Norma is the manager of an automotive parts store and she earns gross income of $2,250 every two weeks. The couple has...
-
One way to determine the number of acidic hydrogens in a molecule is to treat the compound with NaOD in D20, isolate the product, and determine its molecular weight by mass spectrometry. For example,...
-
Treatment of 1-phenyl-2-propenone with a strong base such as LDA does not yield an anion, even though it contains a hydrogen on the carbon atom next to the carbonyl group.Explain....
-
Rollie has a credit card with a credit limit of $4,000. He made the following purchases: $425.36, $358.33, $377.11, and $90.20. What is Rollies available credit?
-
What are the advantages of taking a company public? What are some of the benefits of staying private? Why would a borrower be attracted to lease financing? 2. Is preferred stock more like bonds or...
-
Imagine an investor who purchased a call option with a strike price of 100 for 20. The value of the underlying asset on the date when the option expires is equal to 85. What is the total payoff to...
-
Hanna has a mortgage of $250,000 through the TD Canada Trust for a vacation property. The mortgage is repaid by end of month payments with an interest rate of 5.6% compounded monthly for a term of 2...
-
Reliable Motors shares are expected to pay dividends of $1.35, $1.05, and $1.70 at the end of each of the next three years, respectively. The investor expects the price of the shares at the end of...
-
If the risk-free rate is 5%, the expected market risk premium (rm- rf ) is 10%, the firm has no debt, the equity beta of the firm is 2, the first dividend paid at t=1 is 11, and annual dividends...
-
What percentage of the worlds glacial ice is included in the Antarctic Ice Sheet?
-
Explain the Hawthorne effect.
-
Determine the hybridization about carbon in H 2 CO. a) sp b) sp 2 c) sp 3 d) sp 3 d
-
Two general mechanisms (or various versions of them) for alkene metathesis were originally considered. In the first (pair wise) mechanism, shown in Fig. P18.81, the catalyst brings about cyclobutane...
-
Citaprolam is used as an antidepressant. N(CH,)2 Citaprolam
-
What is dn for each of the following complexes? Pd(PPh3)4
-
Blue-Eyed Beauty Supply is the talk of the town. When Elaine started her business, she had no idea it would evolve to be the number one supplier of cosmetology supply in the region. While her...
-
Mystic Lake Inc. bottles and distributes spring water. On July 9 of the current year, Mystic Lake reacquired 5,700 shares of its common stock at $78 per share. On September 22, Mystic Lake sold 4,000...
-
1. Find the electric flux emanating from a point charge of 6 micro coulombs. 2.An electric field of 500 V/m makes an angle of 30.00 with the surface vector, which has a magnitude of 0.500 m 2 . Find...

Study smarter with the SolutionInn App


