Below are listed the atomic weight, density, and atomic radius for three hypothetical alloys. For each determine
Question:
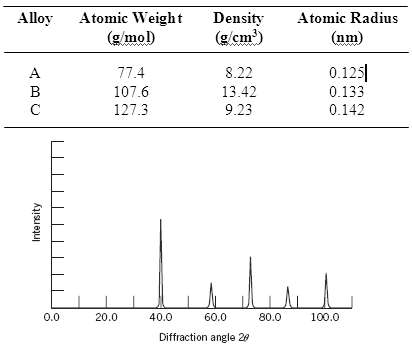
Below are listed the atomic weight, density, and atomic radius for three hypothetical alloys. For each determine whether its crystal structure is FCC, BCC, or simple cubic and then justify your determination. A simple cubic unit cell is shown in Figure 3.24.
Transcribed Image Text:
Alloy Atomic Weight Density (g/cm') Atomic Radius (g/mol) (nm) 0.125| 8.22 77.4 107.6 13.42 0.133 127.3 9.23 0.142 0.0 20.0 40.0 60.0 80.0 100.0 Diffraction angle 20 Intersity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (19 reviews)
For each of these three alloys we need by trial and error to ca...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
The atomic weight, density, and atomic radius for three hypothetical alloys are listed in the following table. For each, determine whether its crystal structure is FCC, BCC, or simple cubic and then...
-
Damocles Company manufactures fine swords. Below are listed the net changes in the companys balance sheet accounts for the past year: The following additional information is available about last...
-
If the pattern shown in Figure 3 is continued indefinitely what fraction of the original square will eventually be painted? Figure 3
-
Rocket Science A rocket has total mass Mi = 360 kg, including 330 kg of fuel and oxidizer. In interstellar space it starts from rest, turns on its engine at time t = 0, and puts out exhaust with...
-
Why is a soft, underinflated football at sea level much firmer when it is taken to a high elevation in the mountains?
-
The Ville de Genve, Switzerland, makes a SFr 400 000 lump-sum budget appropriation to an agency to conduct a counselling programme for drug addicts for a year. All of the appropriation is to be...
-
The velocity components for steady flow through the nozzle shown in Fig. P4.48 are \(u=-V_{0} x / \ell\) and \(v=V_{0}[1+(y / \ell)]\), where \(V_{0}\) and \(\ell\) are constants. Determine the ratio...
-
Saira Morrow operates Dressage Riding Academy, Inc. The academy's primary sources of revenue are riding fees and lesson fees, which are provided on a cash basis. Saira also boards horses for owners,...
-
An air conditioner with cooling capacity of 7 . 3 5 kW consumes 1 . 4 7 kW of electricity during operation. The air conditioner is used to maintain the temperature of a room at 2 0 \ deg C during the...
-
Murphy Delivery Service completed the following transactions during December 2018: Dec. 1 Murphy Delivery Service began operations by receiving $13,000 cash and a truck with a fair value of $9,000...
-
Rhodium has an atomic radius of 0.1345 nm and a density of 12.41 g/cm 3 . Determine whether it has an FCC or BCC crystal structure.
-
The unit cell for tin has tetragonal symmetry, with a and b lattice parameters of 0.583 and 0.318 nm, respectively. If its density, atomic weight, and atomic radius are 7.30 g/cm 3 , 118.69 g/mol,...
-
Great White Trade Mart has recently had lackluster sales. The rate of inventory turnover has dropped, and the merchandise is gathering dust. At the same time, competition has forced Great Whites...
-
Bea and Julia agreed on the sale of Bea's property at the price of P1M. The delivery was agreed to be on March 15, 2020. However, on March 15, 2020, Bea's property is still mortgaged to a bank. What...
-
Suppose the following information is related to legal merger: The combinor issued 10000 common shares with $10 par value and $20 FMV to acquire all the voting common stocks of combinee. The business...
-
The client has provided you with the calculations below, and asked you to determine whether an investment in sector (X) is a profitable investment. We ask you to answer this question through the...
-
The store accountant receives an invoice for $1,400.00 at list price. plus a 554 freight bill with trade discounts of 20 and 10. The in voice is paid on time and earns an additional 2% cash discomm....
-
Write a C program that copies an array A[], inside another array B] (of the same size). The odd elements of A should be at the beginning of B; then the even elements should follow. Do not sort any of...
-
If all eigenvalues of a matrix are real, then the matrix is symmetric. (True/False)
-
How can a promoter avoid personal liability for pre-incorporation contracts?
-
Explain why L 100 R 100 for (x) = x 2 on [3, 7].
-
For each of the following stacking sequences found in FCC metals, cite the type of planar defect that exists: (a) . . . A B C A B C B A C B A . . . (b) . . . A B C A B C B C A B C . . . Now, copy the...
-
(a)Using the intercept method, determine the average grain size, in millimeters, of the specimen whose microstructure is shown in Figure 4.14(b); use at least seven straight-line segments. (b)...
-
(a) Employing the intercept technique, determine the average grain size for the steel specimen whose microstructure is shown in Figure 9.25(a); use at least seven straight-line segments. (b) Estimate...
-
Given the matrix A -3 0 -14 3 4 6 9115 7701 a) Determine all solutions of the homogeneous system Ax = 0. b) Determine if the columns of A span R.
-
Summarize each data source and include them for substance abuse and alcohol in the military. Analyze each data source for substance abuse and alcohol in the military for trustworthiness and accuracy....
-
On January 1, 2023, Holland Corporation paid $7 per share to a group of Zeeland Corporation shareholders to acquire 60,000 shares of Zeeland's outstanding voting stock, representing a 60 percent...

Study smarter with the SolutionInn App


