Below is a unit cell for a hypothetical metal. (a) To which crystal system does this unit
Question:
Below is a unit cell for a hypothetical metal.
(a) To which crystal system does this unit cell belong?
(b) What would this crystal structure be called?
(c) Calculate the density of the material, given that its atomic weight is 141 g/mol.
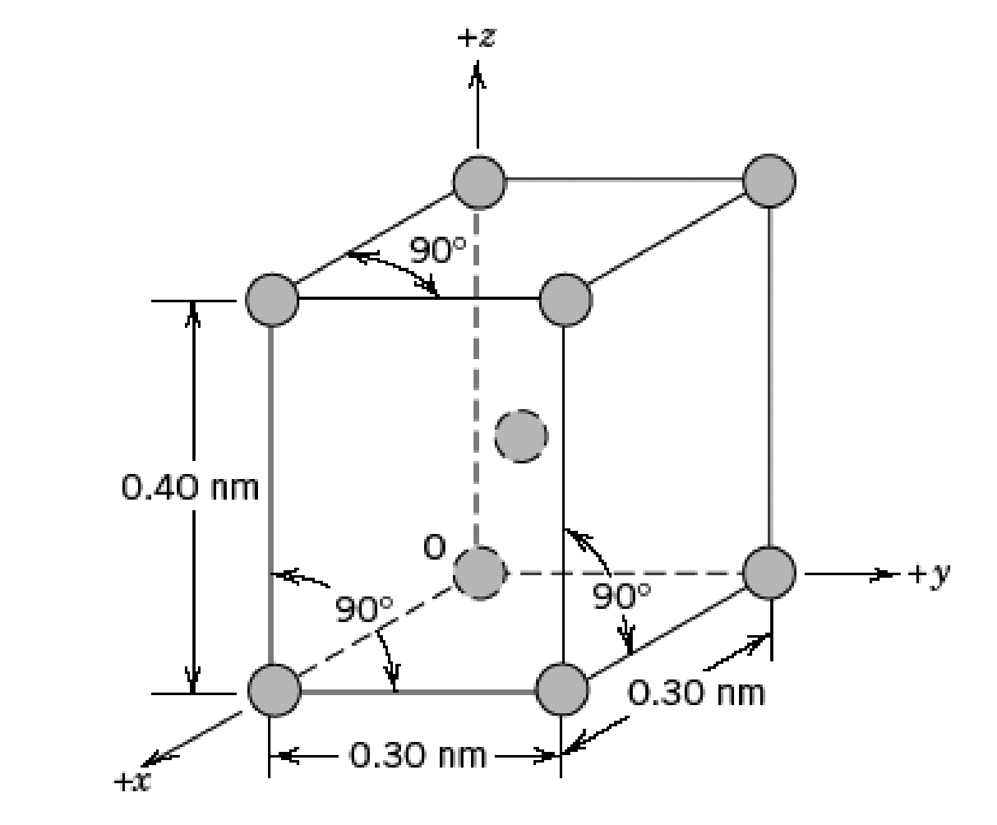
Transcribed Image Text:
+z 90°i 0.40 nm +y 90° 90° 0.30 nm 0.30 nm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a The unit cell shown in the problem statement belongs to the tetragona...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Below is a unit cell for a hypothetical metal. (a) To which crystal system does this unit cell belong? (b) What would this crystal structure be called? (c) Calculate the density of the material,...
-
The accompanying figure shows a unit cell for a hypothetical metal. (a) To which crystal system does this unit cell belong? (b) What would this crystal structure be called? (c) Calculate the density...
-
Calculate the density of hydrogen sulfide gas, H2S, at 56oC and 967 mmHg. Obtain the density in grams per liter.
-
SAE 10 oil at 20C flows in a vertical pipe of diameter 2.5 cm. It is found that the pressure is constant throughout the fluid. What is the oil flow rate in m3/h? Is the flow up or down?
-
Push a pin through a small card and place it in the hole of a thread spool. Try to blow the card from the spool by blowing through the hole, as Evan Jones does in one of the chapter-opening photos....
-
Overloon BV is a repair-service company specialising in the rapid repair of photocopying machines. Each of its 10 clients pays a fixed monthly service fee (based on the type of photocopying machines...
-
As is indicated in Fig. P4.42, the speed of exhaust in a car's exhaust pipe varies in time and distance because of the periodic nature of the engine's operation and the damping effect with distance...
-
For the year ended December 31, 2012, the job cost sheets of Dosey Company contained the following data. Other data: 1. Raw materials inventory totaled $20,000 on January 1. During the year, $100,000...
-
Problem #3: A steady free jet of water exits a 0.5 inch diameter nozzle with uniform velocity V (point 1). The water jet impinges on a flat plate having a weight of 5 lbf and hinged at the left edge...
-
Ashley runs a small business in Boulder, Colorado, that makes snow skis. She expects the business to grow substantially over the next three years. Because she is concerned about product liability and...
-
Rhenium has an HCP crystal structure, an atomic radius of 0.137 nm, and a c/a ratio of 1.615. Compute the volume of the unit cell for Re.
-
Sketch a unit cell for the body-centered orthorhombic crystal structure.
-
You may have seen the mall interviewer, clipboard in hand, approaching people passing by. Explain why even a large sample of mall shoppers would not provide a trustworthy estimate of the current...
-
What actions should the Fed take if it believes the economy is about to experience a high rate of inflation? Now, let's assume you are the President of the Fed and you have to make certain decisions...
-
(a) The liabilities of Pharoah Company are $98,000. Owner's capital is $154,000; drawings are $90,000; revenues, $441,000; and expenses, $323,000. What is the amount of Pharoah Company's total...
-
A model rocket is launched straight upward. Its altitude y as a function of time is given by y = bt -ct2, where b = 82 m/s, c = 4.9 m/s2, t is the time in seconds, and y is in meters, (a) Use...
-
Your company (Amazon, Inc.) would like a presentation of your findings on maximizing profit, pricing decisions, and strategic cooperation at the quarterly board of directors meeting. You will outline...
-
Google has scanned material from over 20 million books so that its search engine can inform users using key word searches about these books. Discuss the legality of this project.
-
Assuming general initial conditions, express the system model in a. Configuration form. b. Standard, second-order matrix form. \(\left\{\begin{array}{l}m \ddot{x}_{1}+k x_{1}-\frac{1}{2}...
-
Wholesalers Ltd. deals in the sale of foodstuffs to retailers. Owing to economic depression, the firm intends to relax its credit policy to boost productivity and sales. The firms current credit...
-
Compute an area function A(x) of (x) with lower limit a. Then, to verify the FTC II inverse relationship, compute A (x) and show that it equals (x). f(x) = x - sinx, a=0
-
Nitrogen from a gaseous phase is to be diffused into pure iron at 700C. If the surface concentration is maintained at 0.1 wt% N, what will be the concentration 1 mm from the surface after 10 h? The...
-
Consider a diffusion couple composed of two semi-infinite solids of the same metal, and that each side of the diffusion couple has a different concentration of the same elemental impurity;...
-
For a steel alloy it has been determined that a carburizing heat treatment of 10-h duration will raise the carbon concentration to 0.45 wt% at a point 2.5 mm from the surface. Estimate the time...
-
Lorenzo Company applies overhead to jobs on the basis of direct materials cost. At year - end, the Work in Process Inventory account shows the following. Work in Process Inventory Date Explanation...
-
T-bill with a $10,000 par has 174 days until maturity. The T-bill has a bank discount bid quote of 2.208% and ask quote of 2.198%. What is the return on the investment in this T-bill if the trader...
-
How much does the company pay in Dividends in 2023 when running the Base Case Drivers?

Study smarter with the SolutionInn App


