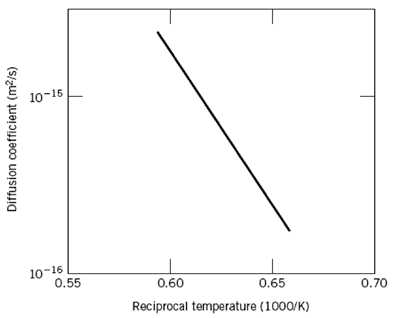
Below is shown a plot of the logarithm (to the base 10) of the diffusion coefficient versus
Question:
Below is shown a plot of the logarithm (to the base 10) of the diffusion coefficient versus reciprocal of the absolute temperature, for the diffusion of iron in chromium. Determine values for the activation energy and preexponential.
Transcribed Image Text:
10-15 10-16 0.55 0.60 0.65 0.70 Reciprocal temperature (1000/K) Diffusion coefficient (m²/s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
This problem asks us to determine the values of Q d and D 0 for the diffusion of Fe in Cr from ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
The accompanying figure shows a plot of the logarithm (to the base 10) of the diffusion coefficient versus reciprocal of the absolute temperature for the diffusion of gold in silver. Determine values...
-
The accompanying figure shows a plot of the logarithm (to the base 10) of the diffusion coefficient versus reciprocal of the absolute temperature for the diffusion of vanadium in molybdenum....
-
A plot of the infiltration curve obtained using Horton's equation is shown in Fig. P2-33. Prove that k = (f0 - fc)/F' if the area between the curve and the fc line. Find the area by integration over...
-
Two bars AB and BC of negligible weight are attached to a single spring of constant k that is unstretched when the bars are horizontal. Determine the range of values of the magnitude P of two equal...
-
Bruing Company's inventory is subject to shrinkage via evaporation. At the end of the current financial reporting period, the company's inventory had a cost of $100,000. Management estimates that...
-
Holmes Manufacturing is considering a new machine that costs $250,000 and would reduce pretax manufacturing costs by $90,000 annually. Holmes would use the 3-year MACRS method to depreciate the...
-
List and distinguish the three views of structural modeling in the context of an example.
-
Dante's Protein Powder produces a dietary supplement. The standard cost of producing one unit is: Material (1.25 pounds $2.50) ..........................$ 3.13 Labor (1.2 hours $10)...
-
I observed a squirrel running across the roof of the my house. The squirrel attempted to jump to a tree. The poor squirrel had bad luck and fell short of the tree. I am happy to report that the...
-
Given a string, reduce it in such a way that all of its substrings are distinct. To do so, you may delete any characters at any index. What is the minimum number of deletions needed? Note: A...
-
The diffusion coefficients for silver in copper are given at two temperatures: (a) Determine the values of D 0 and Q d . (b) What is the magnitude of D at 875C? T (C) D (m?/s) 650 5.5 x 10-16 900 1.3...
-
Carbon is allowed to diffuse through a steel plate 15 mm thick. The concentrations of carbon at the two faces are 0.65 and 0.30 kg C/m 3 Fe, which are maintained constant. If the preexponential and...
-
Companies that sell groceries over the Internet are called e-grocers. Customers enter their orders, pay by credit card, and receive delivery by truck. A potential e-grocer analyzed the market and...
-
Joe owns a bond which is callable in 3 years. The bond has a 6 percent coupon, pays interest semiannually, has a par value of $1,000, and has a yield to call of 6.3 percent. What is the call premium...
-
Compare and contrast the somatic and autonomic division of the peripheral nervous system.
-
If your proposed grade seems too high or too low, your instructor will review the data available (attendance logs, quiz completion, your assignments, etc.) and may adjust your proposed grade up or...
-
Instructions Skandinaviska Enskilda Banken (SEB), one of northern Europe's largest banking groups, operates in several countries, including Germany, Poland, and Russia. In the past, many of SEB's...
-
Northern Metal Manufacturing (NMM), your company, has four manufacturing plants. Sales have been poor lately due to the weak economy that has resulted from the COVID-19 pandemic. Your manager, Marcia...
-
What is high-frequency trading? Describe a strategy through which a high-frequency trader makes a profit.
-
The overall reaction and equilibrium constant value for a hydrogenoxygen fuel cell at 298 K is 2H 2 (g) + O 2 (g) 2H 2 O(l) K = 1.28 10 83 a. Calculate E cell and G 8 at 298 K for the fuel cell...
-
Find the resistance that must be placed in series with a 25.0- galvanometer having a 50.0-A sensitivity (the same as the one discussed in the text) to allow it to be used as a voltmeter with a 3000-V...
-
Is it possible to temper an oil-quenched 4140 steel cylindrical shaft 12.5 mm (0.5 in.) in diameter so as to give a minimum yield strength of 1000 MPa (145,000 psi) and a minimum ductility of 16%EL?...
-
Copper-rich copper-beryllium alloys are precipitation hardenable. After consulting the portion of the phase diagram (Figure 11.30), do the following: (a) Specify the range of compositions over which...
-
A solution heat-treated 2014 aluminum alloy is to be precipitation hardened to have a minimum tensile strength of 450 MPa (65,250 psi) and a ductility of at least 15%EL. Specify a practical...
-
York's outstanding stock consists of 80,000 shares of cumulative 7.5% preferred stock with a $5 par value and also 200,000 shares of common stock with a $1 par value. During its first four years of...
-
Hudson Community College enrolls students in two departments, Liberal Arts and Sciences. The college also has two service departments, the Library and the Computing Services Department. The usage of...
-
Robert is designing a new Active Directory Domain Services infrastructure for a company called Litware, Inc., which has its headquarters in New York and two additional offices in London and Tokyo....

Study smarter with the SolutionInn App


