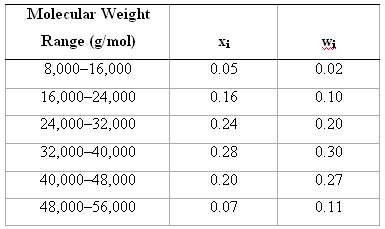
Below, molecular weight data for a polypropylene material are tabulated. Compute (a) The number-average molecular weight, (b)
Question:
Below, molecular weight data for a polypropylene material are tabulated. Compute
(a) The number-average molecular weight,
(b) The weight-average molecular weight, and
(c) The degree of polymerization.
Transcribed Image Text:
Molecular Weight Range (g/mol) Xi Wi 0.02 0.05 8,000-16,000 16,000-24,000 0.16 0.10 24,000-32,000 0.24 0.20 32,000-40,000 0.28 0.30 40,000-48,000 0.20 0.27 48,000-56,000 0.07 0.11
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a From the tabulated data we are asked to compute Mn the numberaverage molecular weight This is carr...View the full answer

Answered By

Joemar Canciller
I teach mathematics to students because I love to share what I have in this field.
I also want to see the students to love math and be fearless in this field.
I've been tutoring these past 2 years and I would like to continue what I've been doing.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Molecular weight data for some polymer are tabulated here. Compute (a) the number-average molecular weight, and (b) the weight-average molecular weight. (c) If it is known that this material's degree...
-
The following table lists molecular weight data for a polytetrafluoroethylene material. Compute the following: (a) The number-average molecular weight (b) The weight-average molecular weight (c) The...
-
Molecular weight data for some polymer are tabulated here. Compute (a) The number-average molecular weight, and (b) The weight-average molecular weight. (c) If it is known that this material's degree...
-
State whether the following statements are true or false. Also, very briefly mention the reason if the statement is false. i. TOFO Surfactants increase the surface tension of water. ii. TOFO The Mach...
-
Do you have enough information to create an implementation plan of AAP. If NOT, how you are going to proceed? Research Good Implementation Plan Research and describe in one paragraph criteria's of...
-
Effect of Helium-Neon Laser Auriculotherapy on Experimental Pain Threshold is the title of an article in the journal Physical Therapy. In this article, laser therapy is discussed as a useful...
-
Repeat the numerical integration in Example 15-5, except use the quadrature formula, Eq. \((9-12)\) Example 5-5 Equation (9-12) A steady-state system of ethanol and water has equimolar...
-
Note 7 to Starbucks 2008 Consolidated Financial Statements presents information about equity method (minority, active) investments. Required a. Starbucks 2008 net income is $315.5 million, and its...
-
In Reflection #1, you considered the fact that it is difficult to understand the morals and ethics of others if we do not truly understand ourselves. Do you feel you have come to know yourself better...
-
Compute the cost of not taking the following trade discounts: (Use 365 days in a year. Round the final answers to 2 decimal places.) a. 2/17, net 40. Cost of lost discount ____% b. 2/17, net 40. Cost...
-
(a) Compute the repeat unit molecular weight of polystyrene. (b) Compute the number-average molecular weight for a polystyrene for which the degree of polymerization is 25,000.
-
Is it possible to have a poly (methyl methacrylate) homopolymer with the following molecular weight data and a of polymerization of 527? Why or why not? Molecular Weight Range (g/mol) Wi Xi...
-
Gaseous A absorbs and reacts with B in liquid according to in a packed bed under conditions where At a point in the reactor where p A = 100 Pa and C B = 100 mol/m 3 liquid (a) Calculate the rate of...
-
5. Two point-like charges are placed at the corners of an equilateral triangle as shown in the figure, a = 38.0 cm. Find the magnitude of the electric field at the top corner. Let q = +1.50 C, and 92...
-
How do contemporary theories of emotional intelligence, such as Mayer and Salovey's model and Goleman's framework, integrate insights from neuroscience, affective psychology, and social cognition to...
-
The summarized income and expense statement for the Dish N' Dat Company product line for the month of March is as follows: Sales Dish N' Dut Company Product-Line Income Statement For the Month Ended...
-
4) Find the magnitude and direction of the net force of the three force vectors , B, and &, shown in the figure. These vectors have the following magnitudes: A = 5.0 N, B = 7.9 N, and C = 8.0 N. What...
-
2.1. Annxn Hilbert matrix, A, is defined by aij=1/(i+j-1) for i, j = 1, 2, ..., n Find the inverse of A and the inverse of ATA for n = 5. Then, noting that (ATA)=A (A-1)T find the inverse of ATA...
-
.. The mother gave birth to the child (Cheryl) on August 29, 1993. In November, 1993, the [Department of Revenue (department)] filed a complaint in the Probate and Family Court against the father on...
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
Chemical species are said to be isoelectronic if they have the same Lewis structure (regardless of charge). Consider these ions and write a Lewis structure for a neutral molecule that is...
-
What properties make copper attractive for cold-working processes?
-
What are some of the limiting properties of copper that might restrict its area of application?
-
What are the two primary types of commercially pure copper and how do they differ?
-
Ruby is 25 and has a good job at a biotechnology company. She currently has $11,000 in an IRA, an important part of her retirement nest egg. She believes her IRA will grow at an annual rate of 9...
-
Susan has a 25-year home mortgage of $210,098 at 4.75% interest and will pay $1,891 annual insurance premium. Her annual property tax will be $2,174. Find her monthly PITI payment. Click the icon to...
-
If a foreign country's consumers tend to only purchase products that are produced locally, the least effective strategy for a U . S . firm is to: a . develop a subsidiary ( under the U . S . name )...

Study smarter with the SolutionInn App


