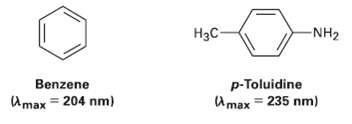
Benzene has an ultraviolet absorption at ? max = 204mm, and para-toluidine has ? max = 235nm.
Question:
Benzene has an ultraviolet absorption at ? max = 204mm, and para-toluidine has ? max = 235nm. How do you account for this difference?
Transcribed Image Text:
-NH2 Нас p-Toluidine U max = 235 nm) Benzene Amax = 204 nm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Double bonds can be conjugated not only with other multiple bonds but also w...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How do you account for Rubbermaids inability to improve its delivery service to Wal-Mart? What factors do you see as contributing to this on-going deficiency?
-
How do you account for Disney management erring so badly, both at the beginning and even for years afterwards? Any suggestions?
-
How do you account for financial losses in order to maintain quality customer service, for example, a restaurant that gives a free meal to an upset customer or a company that gives repeat customers a...
-
Gothic Kings Ltd. Is a 100% owned subsidiary of Hadrian Inc. Gothic has been profitable in the past but incurred a loss for the year ended December 31, 20X3. Hadrian has indicated that if Gothic...
-
Do you think the company's pay structure was better suited to its objectives before or after the reduction in pay level? Why?
-
Explain why it is not advantageous to use more precise rating/ranking scales.
-
Should every board have directors and officers insurance? Why or why not?
-
Norton Company produces two products (Juno and Hera) that use the same material input. Juno uses two pounds of the material for every unit produced, and Hera uses five pounds. Currently, Norton has...
-
A company is switching over to a new system for creating and maintaining employee related records and the employees across the board are unhappy about it . They are not showing up to any trainings...
-
A. The Sunsoria Company employs a job-order pricing method and uses a predetermined overhead rate to apply manufacturing overhead to Work in Process inventory. In the current month, the business had...
-
The double bond of an enamine (alkene amine) is much more nucleophilic than a typical alkene double bond. Assuming that the nitrogen atom in an enamine is sp2-hybridized, draw an orbital picture of...
-
What product would you expect to obtain from a nucleophilic substitution reaction of (S)-2-bromohexane with acetate ion, CH3CO2 ? Assume that inversion of configuration occurs, and show the stereo...
-
Explain the significance of the Clapeyron equation and of the ClausiusClapeyron equation.
-
As a chief financial officer (CFO), which company between Jetblue Airway and Delta do you feel is in better shape, from the standpoint of a manager? Why? (Show several ratios/trends/other pieces of...
-
Consider a computer that uses 8 bits to represent floating-point numbers, 1 bit for the sign s, 3 bits for the exponent c (c =e +3), and 4 bits for fractional part f. In terms of s, e, and f, the...
-
Initially, American Airlines is the only carrier for the New York to Los Angeles route in the U.S. market. Suppose American faces a linear market demand curve: P = 1,140-2Q Where P is price in...
-
The following information is from ABC Company's general ledger: Beginning and ending inventories, respectively, for raw materials were $12,000 and $15,000 and for work in process were $30,000 and...
-
Which framework should be used to Evaluate each of the three companies' plans for entering Ethiopia. What are the most salient risks for each and what might they do to mitigate those risks?
-
Consider the following mutually exclusive projects: Amounts are expressed in thousands of dollars and correspond to the cash flows expected to occur at the end of each period. Period 0 is today,...
-
Why do CPA firms sometimes use a combination of positive and negative confirmations on the same audit?
-
If the solventsolute interactions in a mixture are comparable in strength to the solventsolvent interactions and the solutesolute interactions, what can you conclude about solution formation in this...
-
Show how to synthesize the following amines from the indicated starting materials by reductive amination. (a) Benzylmethylamine from benzaldehyde (b) N-benzylpiperidine from piperidine (c)...
-
Show how to synthesize the following amines from the indicated starting materials by acylation-reduction. (a) N-butylpiperidine from piperidine (b) N-benzylaniline from aniline
-
Addition of one equivalent of ammonia to 1-bromoheptane gives a mixture of heptan-1-amine, some dialkylamine, some trialkylamine, and even some tetraalkylammonium bromide. (a) Give a mechanism to...
-
ELA, Inc. will pay a $ 0 . 6 0 dividend today. The dividend is expected to triple in the first year and then double in the second year. After that the dividend will grow at a constant annual rate of...
-
1 Skipped An investor is considering the purchase of either an IO or PO strip from a CMO offering. The portion of the mortgage pool backing this tranche consists of $1.18 million in mortgages with a...
-
A zero coupon bond has a par value of RM1,000 and matures in 20 years. Investors require a 10% annual return on these bonds. For what price should the bond sell? (note, zero coupon bonds do not pay...

Study smarter with the SolutionInn App


