Calculate Ksp for iron(II) sulfide given the following data: Es. cell
Question:
Calculate Ksp for iron(II) sulfide given the following data:
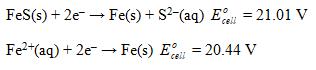
Transcribed Image Text:
Es. cell
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
The K sp reaction is FeSsFe 2 aq S 2 aq K K sp Manipulate the give...View the full answer

Answered By

Shivani Dubey
Hello guys, I am very good with the Advance mathematics, Calculus, Number theory, Algebra, Linear algebra, statistics and almost all topics of mathematics. I used to give individual tutoring students in offline mode but now want to help students worldwide so I am joining here. I can also give solutions in various coding languages of mathematics and software like mathematica, R, Latex, Matlab, Statistica, etc.
feel free to ask any doubt regarding mathematics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Given the following data about the economy: a. Calculate aggregate output (GDP) and aggregate income. b. Compare the two calculations in a. Why are they not precisely equal? c. Calculate GNP. d....
-
Given the following data, calculate product cost per unit under variable costing. Direct labor $8 per unit Direct materials $3 per unit Overhead Total variable overhead $30,000 Total fixed overhead...
-
Given the following data for Profiteers, Inc., and the corresponding industry averages, perform a trend analysis of the return on investment and the return on stockholders equity. Plot the data and...
-
Suppose that Canada produces two goods: lumber and fish. It has 18 million workers, each of whom can cut 10 feet of lumber or catch 20 fish each day. a. What is the maximum amount of lumber Canada...
-
You have $200 and are thinking about betting on the Big Game next Saturday. Your team, the Golden Boars, are scheduled to play their traditional rivals the Robber Barons. It appears that the going...
-
Is a corporation that participates in a public debate subject to liability for factual inaccuracies?
-
Table 1.2 illustrates a possible scenario in a trade on gold futures. On day 1 , when the gold futures price is \(\$ 1350\) per ounce, we enter a long position for two contracts, whose unit size is...
-
Jimmy Olson died on June 15, 2011, at the age of 75, after a brief illness. Jimmy is survived by his wife, Lois, and two adult sons, Clark and Kent. Jimmy left a valid will, requesting that Clark...
-
(a) (b) In a liberalised economy, market forces of demand and supply dictate the interest rates and exchange rates in the market. With reference to the above statement, analyse two justifications why...
-
Paper purchased 12,000 of Sands 30,000 shares on 10/1/20X0 for $36,000. At 1/1/20X0, Sand had Common Stock of $30,000 and Retained Earnings of $50,000. During the 12 months ended 12/31/20X0, Sand...
-
For the following half reaction, E cell = 22.07 V: AlF 6 3 + 3e - Al + 6F 2 Using data from Table 11.1, calculate the equilibrium constant at 25 o C for the reaction Al 3+ (aq) 1 6F 2 (aq) AlF 6 3...
-
CuI + e Cu + I2
-
Ogden issued a $70,000, 12%, 10-year bond payable at 90 on January 1, 2016. Requirements 1. Journalize the issuance of the bond payable on January 1, 2016. 2. Journalize the payment of semiannual...
-
Whitman Company Income Statement Sales (42,000 units x $41.60 per unit) Cost of goods sold (42,000 units x $21 per unit) Gross margin Selling and administrative expenses Net operating income $...
-
Mini-Case: Tax Deductions and Tax Credits Ted Burns Your investment client, Ted Burns, is a 55-year-old service manager for a car dealership. Ted has asked you for advice on preparing his 2017 tax...
-
Valence Electronics has 231 million shares outstanding. It expects earnings at the end of the year of $810 million. Valence pays out 40% of its earnings in total - 15% paid out as dividends and 25%...
-
Explain two expected differences in behaviors between members of individualist and collectivist cultures with Examples
-
During the current year, Ron and Anne sold the following assets: ( Use the dividends and capital gains tax rates and tax rate schedules. ) Capital Asset Market Value Tax Basis Holding Period L stock...
-
The shareholders equity section of CSR Limiteds 2017 balance sheet is shown below. 1. Provide a reason for the change in share capital. 2. Explain what is meant by non-controlling interests. 3....
-
Diamond Walker sells homemade knit scarves for $25 each at local craft shows. Her contribution margin ratio is 60%. Currently, the craft show entrance fees cost Diamond $1,500 per year. The craft...
-
In each of the following scenarios, explain the changes in either supply or demand that would result. If the initial equilibrium price were yet to change, indicate whether a surplus or a shortage...
-
According to a 2016 article in the Wall Street Journal, After years of relative equilibrium, the job market for nurses is heating up in many markets, driving up wages and sign-on bonuses for the...
-
Suppose the supply of green tea increases; why is it that equilibrium price and equilibrium quantity move in opposite directions?
-
How do I view vector and bitmap images in a VR headset?
-
The radius of a solid conducting sphere is R = 14.0 cm and it has a uniformly distributed charge of 30.5 nC on its surface. The center of the sphere is at the origin of the xy coordinate system. The...
-
What is the best file format for digital images, such as photos and scans?

Study smarter with the SolutionInn App


