Calculate product rates and compositions, stage temperatures, and interstage vapor and liquid flow rates and compositions for
Question:
Calculate product rates and compositions, stage temperatures, and interstage vapor and liquid flow rates and compositions for an absorber having four equilibrium stages with the specifications in figure. Assume the oil isnC10.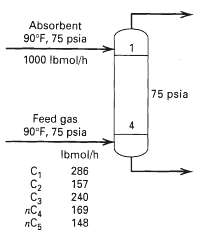
Transcribed Image Text:
Absorbent 90°F, 75 psia 1000 Ibmol/h 75 psia Feed gas 90°F, 75 psia 4 Ibmol/h 286 C2 C3 пС 157 240 169 148 తొలోలొలె
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
As shown below when the gas feed was flashed it was found to contain 72 mol liquid The c...View the full answer

Answered By

Diksha Bhasin
I have been taking online teaching classes from past 5 years, i.e.2013-2019 for students from classes 1st-10th. I also take online and home tuitions for classes 11th and 12th for subjects – Business Studies and Economics from past 3 years, i.e. from 2016-2019. I am eligible for tutoring Commerce graduates and post graduates. I am a responsible for staying in contact with my students and maintaining a high passing rate.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Calculate product compositions, stage temperatures interstage vapor and liquid flow rates and compositions, reboiler duty, and condenser duty for the following distillation-column specifications Feed...
-
Calculate the equilibrium constant for the reaction O2 2O at temperatures of 537 R and 10 000 R.
-
Calculate the IRR of each of the four stages of the cost reduction proposal in Problem 2 of Exercise 16.2. Based on the IRR investment criterion, which stages should be approved at a 14% cost of...
-
In July 2009, Malcolm bought a piece of land for 40,000. In June 2016 he sold part of the land for 11,000. This disposal was not caused by a compulsory purchase and was his only disposal of land in...
-
The following information is for Kon Inc. for the year ended December 31, 2012: Common shares, Jan. 1..................................$20,000 Common shares issued during year..............10,000...
-
Phil and Marcy have been married for a number of years. Marcy is very wealthy, but Phil is not. In fact, Phil, who has only $10,000 of property, is very ill, and his doctor believes that he probably...
-
As the in-charge auditor, you begin your field work to examine the December 31 financial statements of a client on January 5, knowing that you must leave temporarily for another engagement on January...
-
Valuation Vandells free cash flow (FCF0) is $2 million per year and is expected to grow at a constant rate of 5% a year: its beta is 1.4. What is the value of Vandells operations? If Vandell has...
-
The Institute of Business Ethics states that its role is to: "... raise public awareness of the importance of doing business ethically... and to help organisations to strengthen their ethics culture...
-
Read the Case of Goodwill Impairment at Jackson Enterprises . After reading the case study, answer the questions provided. Financial reporting personnel at Jackson Enterprises (JE) are in the process...
-
An absorber is to be designed for a pressure of 75 psia to handle 2,000 lbmol/h of gas at 60?F having the following composition.The absorbent is an oil, which can be treated as a pure component...
-
In Example 10.4, temperatures of the gas and oil, as they pass through the absorber, increase substantially. This limits the extent of absorption. Repeat the calculations with a heat exchanger that...
-
Refer to the information in P1217. Assume the U.S. dollar is the functional currency, not the krone. Required a. Prepare a schedule remeasuring the trial balance from Norwegian kroner into U.S....
-
Find the conditional \(p d f\) for one discrete random variable given the value of another and their joint \(p d f\).
-
Explain the logic of a statistical test, including why it is important that a test statistic has a known probability distribution if the null hypothesis is true.
-
Explain the difference between one-tail and two-tail tests. Explain, intuitively, how to choose the rejection region for a one-tail test.
-
Explain how you would go about choosing a functional form and deciding that a functional form is adequate.
-
A big question in the United States, a question of "cause and effect," is whether mandatory health care will really make Americans healthier. What is the role of hypothesis testing in such an...
-
The partners are to change their profit ratios as shown: They decide to bring in a goodwill amount of 150,000 on the change. The last statement of financial position before any element of goodwill...
-
1. True or False. Pitfalls to consider in a statistical test include nonrandom samples, small sample size, and lack of causal links. 2. Because 25 percent of the students in my morning statistics...
-
Identify three self-regulating agencies and explain which industries they regulate. Speculate as to why an industry would self- regulate.
-
Derive (15-44) and use it to solve the following problem. Sulfate ion is to be removed from 60 L of water by exchanging it with chloride ion on 1 L of a strong-base resin with relative molar...
-
For mass transfer outside a single spherical particle that is not close to a wall or other particles, what is the smallest value of the Sherwood number? What is the basis for this value?
-
Within a porous particle, why are mass and heat transfer not analogous?
-
Differentiate between a scalar value and a vector value.
-
Now that the supplier of the nori has been chosen and the transportation method selected, you turn your attention to the downstream side of the supply chain -- the marketing (distribution) channels....
-
Mandy Murphy, owner of Murphy and Co. (Saint John, New Brunswick), gave the following list of assets and liabilities to a public accountant and asked him to prepare a balance sheet for the company as...

Study smarter with the SolutionInn App


