Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c)
Question:
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds:
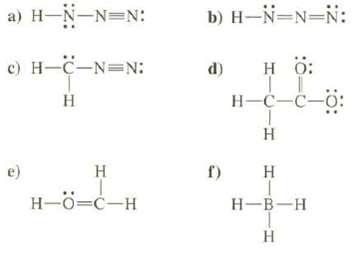
Transcribed Image Text:
a) H-N-N=N: c) H-C-N=N: H e) H H-Ö=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (19 reviews)
1 1 0 a HNNN 0 H ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Calculate the formal charges on the atoms shown inred. (a) (CH3)2OBF3 (b) H2C-NEN: (c) H2C=N=N: (d) :=-: (e) H (f) - CH
-
Calculate the formal charges on each of the atoms, except hydrogen's, of the molecules and determine the total charge of the species: a) H H-C-0_H d) H H C=N=N: b) H H-C-0: f) SF: : | H-NC-C- ...
-
Verify that the formal charges on nitrogen in ammonium ion and boron in borohydride ion are as shown.
-
A certain radioactive isotope is a by - product of some nuclear reactors. Due to an explosion, a nuclear reactor experiences a massive leak of this radioactive isotope. Fortunately, the isotope has a...
-
Explain the meaning of supply chain and provide an example to illustrate your answer. Your example should focus on a particular industry and involve at least one answer customer.
-
July sales for Melodic Kortholt, Ltd., average 1 = $9,500 with 12 5 $1,250. August sales average 2 = $7,400 with 22 = $1,425. September sales average 3 $8,600 with 32 = $1,610. (a) Find the mean and...
-
Calculate NewWorlds price-earnings ratio, before and after the takeover described in Example 22.2.
-
The Laurel Co. is owned and operated by Paul Laurel. The following is an excerpt from a conversation between Paul Laurel and Maria Fuller, the chief accountant for Laurel Co. Paul: Maria, Ive got a...
-
19 20 Assertion A compass needle is placed near a current carrying wire. The deflection of the compass needle decreases when the magnitude of the current in the wire is increased. Reason The strength...
-
A teacher has given four tests to a class of five students and stored the results in the following matrix Discuss methods of matrix multiplication that the teacher can use to obtain the information...
-
Show Lewis structures for these compounds: (a) CH5N (b) C 2 H 5 Cl (c) N 2 (d) CH 2 S (e) C 2 H 3 F (f) CH 4 S
-
Explain which of the two following structures would be more stable. Explain whether they represent isomers or are resonance structures. HIN: H N-H H :0: N-H HIN
-
If John wants to retire with \(\$ 10,000\) per month, how much principal is necessary to generate this amount of monthly income if the interest rate is \(4 \%\) ?
-
Given that the remainder is 15 , the quotient is 2x^(2)+3x-7, and the divisor is x+2, determine the dividend.
-
Carlos purchased a new business asset (five-year property) on February 10, 2023, at a cost of $100,000. Carlos elected to expense $10,000 of the cost immediately under 179 and the remainder using...
-
Charles owns a mobile home He would like to place it in thr shades woods mobile home park and signs a lease allowing him to park the mobile home on a particular lot this is know as?
-
Assume the following information from a schedule of cost of goods manufactured: Cost of goods manufactured $ 158,000 Beginning work in process inventory $ 30,000 Direct materials used in production $...
-
which forecasting methods are most suitable (out of holts exponential smoothing method, simple exponential smoothing and holts winter exponential smoothing) for the graphs seen below? graph 1...
-
A system is represented by equation \(\dot{\mathbf{x}}=\mathbf{A x}\) where \[A=\left[\begin{array}{ccc}0 & 1 & 0 \\0 & 0 & 1 \\-1 & -c & -2\end{array}ight]\] Find the range of \(c\) where the system...
-
Do public and private companies follow the same set of accounting rules? Explain.
-
Use the laws of logarithms to evaluate log 2 80 log 2 5.
-
Divalent carbon species called carbenes care capable of fleeting existence. For example, methylene: CH2, is the simplest carbene. The two unshared electrons in methylene can be either spin-paired in...
-
There are two different substances with the formula C4H10. Draw both, and tell how they differ.
-
There are two different substances with the formula C3H6. Draw both, and tell how they differ.
-
What are the key segments in the premium dog market? What key characteristics do customers in the premium dog segment share? How does this influence their buying process for their dogs? Explain the...
-
I have seen an example of an emerging approach that has been successful in the commercial world. Competition from other businesses operating in the same industry was fierce for the company I worked...
-
give me some Marketing strategies for a Trade School to Engage Students How make those strategies works? Also, ideas to Recruit Students for Trade School with Modern Marketing Tactics

Study smarter with the SolutionInn App


