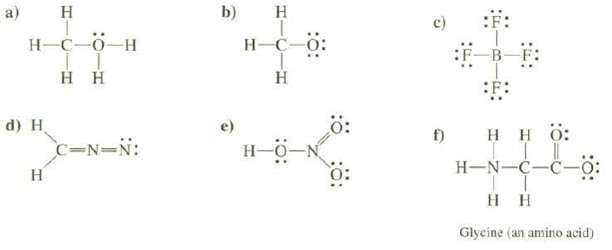
Calculate the formal charges on each of the atoms, except hydrogen's, of the molecules and determine the
Question:
Calculate the formal charges on each of the atoms, except hydrogen's, of the molecules and determine the total charge of the species:
Transcribed Image Text:
a) H H-C-0_H Η Η d) H H C=N=N: b) H H-C-0: Η Ξ f) SF: Η Η Ο: | H-NC-C- Τ Η Η Glycine (an amino acid)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
The number of valence electrons is the same as the group number of the atom Unshared electron...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c) H-C-N=N: H e) H H-=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
-
Calculate the formal charges on the atoms shown inred. (a) (CH3)2OBF3 (b) H2C-NEN: (c) H2C=N=N: (d) :=-: (e) H (f) - CH
-
Verify that the formal charges on nitrogen in ammonium ion and boron in borohydride ion are as shown.
-
Montage Pty Limited (Montage) is a resident private company and is not a base rate entity. Which of the following transactions would result in a debit entry to Montage's franking account? Payment of...
-
Sweety Tea (ST) manufactures a range of designer T-shirts, which it produces to order for major department stores. On to March the sales department at ST received an order from retail company, Legend...
-
M&Ms are blended in a ratio of 13 percent brown, 14 percent yellow, 13 percent red, 24 per- cent blue, 20 percent orange, and 16 percent green. Suppose you choose a sample of two M&Ms at random from...
-
Dairymaid makes organic yogurt. The only ingredients, milk and bacteria cultures, are added at the very beginning of the fermentation process. At month end, Dairymaid has 100,000 cups of yogurt that...
-
Chester Inc. has identified activity centers to which overhead costs are assigned. The cost pool amounts for these centers and their selected activity drivers for 2013 follow. The companys products...
-
16. A signal containing multiple frequencies is shown in figure A. It is passed through 2 different filters and then outputs are shown in figure B & C. M (A) S (B) sha (C) (a) B corresponds to low...
-
George Jack operates a small machine shop. He manufactures one standard product available from other similar businesses, and he also manufactures products to customer order. His accountant prepared...
-
Using Lewis structures show a balanced equation for the reaction of H2O with HCl.
-
Predict which of the following constitutional isomers for the compound that is formed from one atom each of hydrogen, oxygen, and chlorine is more stable: H-CI-: or H-O-CI:
-
In January 2024, Burrard Shipyards Ltd. (BSL) was awarded a $42-million contract to construct a non-combat hospital ship for the Royal Canadian Navy. The contract was part of the federal governments...
-
Find the LCM of x - 2x - 15 and x - 3x - 18.
-
You own a property on the outskirts of the city and are considering building either a 5.000 square meter apartment block with five units or two single-family homes with 2.000 square meters each. The...
-
Describe the early theory of management. Compare the differences between the theory you have chosen and at least one current approach/theory of management. In your comparison, include a minimum of...
-
Solve the equation. (Enter your answers as a comma-separated list.) 16*-7 = 644-x
-
Compare and Criticize the existing structure of any two named organization of your choice. What difference do you notice in their respective structures after each of them diversify their businesses.
-
In the diagram for Problem 4.1, a PID controller is used in a unit feedback system such that (a) What is the effect of the PID on the stability of this specific system? (b) Find the steady-state...
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
Express ln a + 1/2 ln b as a single logarithm.
-
The following model is a representation of asparatame, C14H18N2O5, known commercially as NutraSweet. Only the connection between atoms is shown; multiple bonds are not indicated. Complete the...
-
How many valence electrons does the each of the following dietary trace elements have? (a) Zinc (b) Iodine (c) Silicon (d) Iron
-
Give the ground-state electron configuration for each of the following elements: (a) Potassium (b) Arsenic (c) Aluminum (d) Germanium
-
Jen and Barry's ice cream shop charges $1.65 for a cone. Variable expenses are $0.31 per cone, and fixed costs total $2,000 per month. A Valentine's Day promotion is being planned for the second week...
-
a. The future value of a $1,050 savings deposit after five years at an annual interest rate of 4 percent. (Round FV factor to 3 decimal places and final answer to 2 decimal places.) Future value b....
-
QuestromT Not complete Marked out of 2.00 ring question Homework - Week 6 - Module 23 Determining Unit Costs, Variance Analysis, and Interpretation Big Dog Company, a manufacturer of dog food,...

Study smarter with the SolutionInn App


