Calculate the number density of free electrons in (a) Ag (? = 10.5 g/cm3) and (b) Au
Question:
Calculate the number density of free electrons in
(a) Ag (? = 10.5 g/cm3) and
(b) Au (? = 19.3 g/cm3), assuming one free electron per atom, and compare your results with the values listed in Table 27-1.
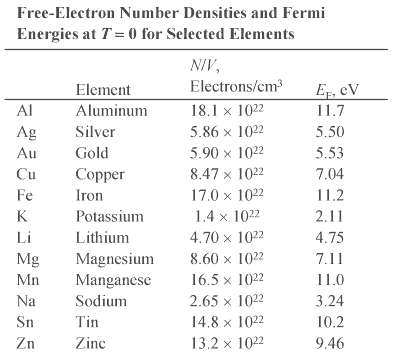
Transcribed Image Text:
Free-Electron Number Densities and Fermi Energies at T= 0 for Selected Elements NIV, Electrons/cm E, cV Element Al Aluminum 18.1 x 1022 11.7 Silver 5.50 Ag 5.86 x 1022 5.53 Au Gold 5.90 x 1022 7.04 Cu Copper 8.47 x 1022 Fe Iron 17.0 x 1022 11.2 K Potassium 1.4 x 1022 2.11 Li Lithium 4.70 x 1022 4.75 8.60 x 1022 Mg Magnesium Manganese 7.11 Mn 16.5 x 1022 11.0 Na Sodium 2.65 x 1022 3.24 14.8 x 1022 Sn Tin 10.2 Zn Zinc 13.2 x 1022 9.46
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
a b n N A M density M molar mass a n Ag 586 10 22 elcm ...View the full answer

Answered By

Saud Ur Rehman
Evaluating manufacturing processes by designing and conducting research programs; applying knowledge of product design, fabrication, assembly, tooling, and materials; conferring with equipment vendors; soliciting observations from operators. Developing manufacturing processes by studying product requirements; researching, designing, modifying, and testing manufacturing methods and equipment; conferring with equipment vendors. Keeping equipment operational by coordinating maintenance and repair services; following manufacturer's instructions and established procedures; requesting special service.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Electricity and Magnetism questions
-
Calculate the number density of free electrons for (a) Mg (? = 1.74 g/cm3) and (b) Zn (? = 7.1 g/cm3), assuming two free electrons per atom, and compare your results with the values listed in Table...
-
The number density of free electrons in copper is 8.47 ?? 1022 electrons per cubic centimeter. If the metal strip in Figure is copper and the current is 10 A, find (a) The drift velocity vd and (b)...
-
In the discussion of free electrons in Section 42.5, we assumed that we could ignore the effects of relativity. This is not a safe assumption if the Fermi energy is greater than about 1/100 mc2 (that...
-
Caine Bottling Corporation is considering the purchase of a new bottling machine. The machine would cost $200,000 and has an estimated useful life of 8 years with zero salvage value . Management...
-
On June 30 the general ledger of Kisling, Inc., had the following balances: Purchases ....................................................................$42,880 Dr. Freight In...
-
Welte Mutual Funds, Inc., is located in New York City. Welte just obtained $100,000 by converting industrial bonds to cash and is now looking for other investment opportunities for these funds. Based...
-
Implement a Bayesian belief network in the programming language of your choice to represent a subject in which you are interested (for example, you might use it to diagnose medical conditions from...
-
Cody Macedo established an insurance agency on January 1 of the current year and completed the following transactions during January: a. Opened a business band account with a deposit of $75,000 in...
-
-S Data concerning a recent period's activity in the Prep Department, the first processing department in a company that uses process costing, appear below: Equivalent units in ending work in process...
-
Shake Shack Incorporated, which began as a hot dog stand in 2001, now has more than 200 locations worldwide. The following is adapted from Shake Shack's financial statements for the quarter ended...
-
(a) Given a mean free path = 0.4 nm and a mean speed vav = 1.17 105 m/s for the current flow in copper at a temperature of 300 K, calculate the classical value for the resistivity of copper. (b)...
-
The density of aluminum is 2.7 g/cm3. How many free electrons are present per aluminum atom?
-
Explain the following analogy: Influence is to leadership, as eggs are to an omelet.
-
The center-to-center distance between a \(200 \mathrm{~g}\) lead sphere and an \(800 \mathrm{~g}\) lead sphere is \(0.120 \mathrm{~m}\). A \(1.00 \mathrm{~g}\) object is placed \(0.0800 \mathrm{~m}\)...
-
Hannah and Daughters is a United States-based graphic design firm. It has a subsidiary in Montenegro that generates foreign EBIT of 30 million euros. The firm has tangible assets overseas with a book...
-
Derive expressions for the orbital speed and energy for a satellite of mass \(m_{\mathrm{s}}\) traveling in a circular orbit of radius \(a\) around a planet of mass \(m_{\mathrm{p}} \gg...
-
Accounts and notes receivable are reported in the current assets section of the statement of financial position at: a. cash (net) realizable value. b. net book value. c. lower-of-cost-or-net...
-
What maximum height above the surface of Earth does an object attain if it is launched upward at \(4.0 \mathrm{~km} / \mathrm{s}\) from the surface?
-
A particle moves with the velocity-versus-time graph shown here. At which labeled point is the magnitude of the acceleration the greatest? A Vx B C D
-
What are the before image (BFIM) and after image (AFIM) of a data item? What is the difference between in-place updating and shadowing, with respect to their handling of BFIM and AFIM?
-
Convert: (a) 4.50 Cal to calories (b) 600.0 Cal to kilojoules (c) 1.000 J to calories (d) 50.0 Cal to joules
-
The two forces F1 and F2 shown in Fig. 4-43a and b (looking down) act on a 27.0-kg object on a frictionless tabletop. If F1 = 10.2N and F2 = 16.0N, find the net force on the object and its...
-
One 3.2-kg pain bucket is hanging by a mass less cord from another 3.5-kg paint bucket, also hanging by a mass-less cord, as shown in Fig.4-44. (a) If the buckets are at rest, what is the tension in...
-
A person pushes a 14.0-kg lawn mower at constant speed with a force of F = 88.0 N directed along the handle, which is at an angle of 45.0o to the horizontal (Fig.4-45). (a) Draw the free-body diagram...
-
The article "SAC Case Tests a Classic Dilemma" describes how certain aspects of the government's insider trading case against SAC Capital resembles the standard prisoner's dilemma game as well as...
-
Lamar's capital balance is $40,960 after admitting Terrell to the partnership by investment. If Lamar's ownership interest is 20% of total partnership capital, what were (1) Terrell's cash investment...
-
+ The cash balances per books for the Jackson Company on November 30, 2002 and December 31, 2003, are $3,625 and $10,967 respectively. The following checks and receipts were recorded on the books for...

Study smarter with the SolutionInn App


