Calculate the theoretical masses of the species in Figure 21-9 and compare your answers with the values
Question:
Calculate the theoretical masses of the species in Figure 21-9 and compare your answers with the values observed in the figure.
Figure 21-9
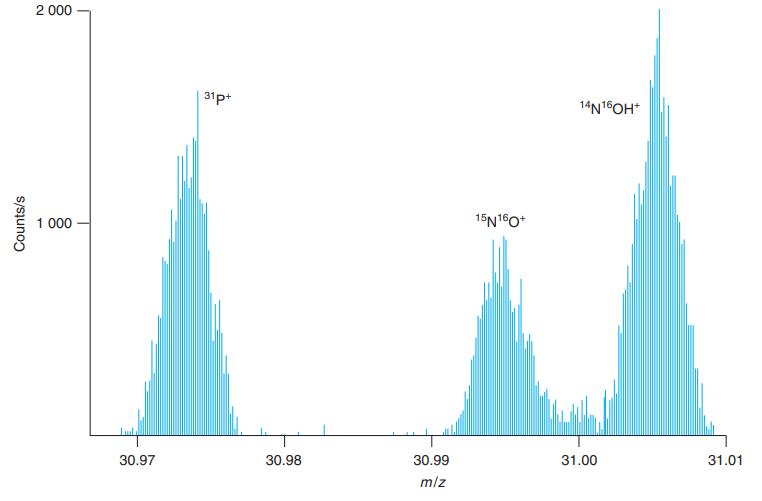
Transcribed Image Text:
2 000 31p+ 14N16OH* 1 000 15N160* 30.97 30.98 30.99 31.00 31.01 m/z Counts/s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
31P 31P e 30973 760000 55 30973 21 observed 309735 To measure mz I enlar...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Calculate the theoretical air-fuel ratio on a mass and mole basis for the combustion of ethanol, C2H5OH.
-
Calculate the theoretical air-fuel ratio on a mass and mole basis for the combustion of ethanol, C2H5OH.
-
Calculate the theoretical density of -Sn. Assume the diamond cubic structure and obtain the atomic radius information from Appendix B.
-
What implications might the difference in initial training provision between the USA and Germany have for the organisation of work and the character of relationships between managers and...
-
Terando Co. began operations on July 1. It uses a perpetual inventory system. During July, the company had the following purchases and sales. Instructions (a) Determine the ending inventory under a...
-
Why is the executive summary an important part of the report?
-
True or False. There is no need for trending of bearings. Bearings have a calculatable performance life, known as the L10 life, and the decision to replace them can be automatically timed by a...
-
Syracuse Beverages Inc. has three plants that make and bottle cola, lemon-lime, and miscellaneous flavored beverages, respectively. The raw materials, labor costs, and automated technology are...
-
Discuss the principles and applications of supercritical fluid extraction (SFE), focusing on the advantages of using supercritical fluids over traditional solvents, and the optimization of extraction...
-
An investment analyst collects data on stocks and notes whether or not dividends were paid and whether or not the stocks increased in price over a given period. Data are presented in the following...
-
The mass of a fragment ion in a high-resolution spectrum is 83.086 5 Da. Which composition, C 5 H 7 O + or C6H + 1 1, better atches the observed mass?
-
Consider the extraction of Mn+ from aqueous solution into organic solution by reaction with protonated ligand, HL: Rewrite Equation 22-13 in terms of Kextraction and express Kextraction in terms of...
-
Design an Ajax style of Web page that would allow a dean at a community college to select part-time instructors. The dean should be able to select a discipline or a course and have the server send an...
-
What are the benefits and risks created by unregulated decentralised finance (DeFi) that is currently seamlessly operating outside of regulatory structures/bodies and expansion into financial...
-
How and why might temperature affect pigment?
-
When designing a new brewery, Laurence needs to balance future capacity with the current cost of construction. Thinking about costs of capital and the time value of money, what do you think her...
-
Does cash flow information not include cash receipts? Please clarify with Example.
-
Decentralised Finance (DeFi), an emerging peer-to-peer financial technology based on blockchain networks, smart contracts and dApps, is intended to remove the control centralised institutions such as...
-
One of the biggest purchases we make in our lifetimes is for a home. Questions that we all ask are these: How much should I spend for a particular home? How many bathrooms are there? How long...
-
Swifty company is a publicly held corporation whose $1 par value stock is actively traded at $30 per share. The company issued 3400 shares of stock to acquire land recently advertised at $93000. When...
-
A 50.0-mL solution containing Ni 2+ and Zn 2+ was treated with 25.0 mL of 0.045 2 M EDTA to bind all the metal. The excess unreacted EDTA required 12.4 mL of 0.012 3 M Mg 2+ for complete reaction. An...
-
Considering just acid-base chemistry, not ion pairing and not activity coefficients, use the systematic treatment of equilibrium to find the pH and concentrations of species in 1.00 L of solution...
-
What is the difference between E and E o for a redox reaction? Which one runs down to 0 when the complete cell comes to equilibrium?
-
A friend owes you $500 in 1 year from now, and then $1000 2 years from now. Both loans are at 5%. If you want to replace both of these loans with ONE PAYMENT NOW, how much would your friend give you ?
-
If the amount of retained earnings at the beginning of the year was $ 3 0 , 0 0 0 , and $ 1 2 , 0 0 0 in dividends is paid during the year, calculate net income for the year.
-
On January 2, 20Y4, Whitworth Company acquired 33% of the outstanding stock of Aloof Company for $330,000. For the year ended December 31, 20Y4, Aloof Company earned income of $86,000 and paid...

Study smarter with the SolutionInn App


