Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves
Question:
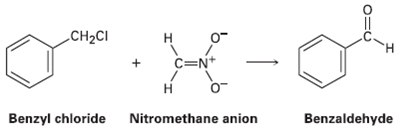
Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves initial conversion of nitro methane into its anion, followed by SN2 reaction of the anion with benzyl chloride and subsequent E2 reaction. Write the mechanism in detail, using curved arrows to indicate the electron flow in each step.
Transcribed Image Text:
CH2CI н C=N+ Н Benzyl chloride Nitromethane anion Benzaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
09100 CH 0 ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using curved arrows to indicate the election flow in each step, show how the base-catalyzed reverse aldol reaction of 4-hydroxy-4-methly-2-pentanone takes place to yield 2 equivalents of acetone.
-
Benzyl bromide is converted into benzaldehyde by heating in dimethyl sulfoxide. Propose a structure for the intermediate, and show the mechanisms of the two steps in thereaction. CH2BR E2 reaction...
-
Show how 4-methylcyclohexyl chloride can be converted to 4- methylcyclohexanecarboxylic acid.
-
IFRS requires the following: (a) Ending inventory is written up and down to net realizable value each reporting period. (b) Ending inventory is written down to net realizable value but cannot be...
-
The model below depicts industry analysis as a funnel that focuses on remote-factor analysis to better understand the impact of factors in the operating environment. Do you find this model...
-
Describe what is meant by mitigation. Explain how the obligation to mitigate damages limits the ability of the victim of a breach to obtain damages.
-
A long straight wire carries a current of magnitude \(I\). Use the Biot-Savart law to derive an expression for the mag. netic field \(\vec{B}\) produced at point \(\mathrm{P}\) a radial distance...
-
Division A offers its product to outside markets for $30. It incurs variable costs of $11 per unit and fixed costs of $75,000 per month based on monthly production of 4,000 units. Division B can...
-
The definition, " ' Chiropractor ' means a person who is a medical quack with no legitimate scientific background," is an example of: Theoretical definition Definition by subclass Persuasive...
-
There is a stock index futures contract maturing in one year. The risk-free rate of interest for borrowing is 3.4% per annum, and the corresponding risk-free rate for lending is 0.7% per annum lower....
-
Rank the following substituted phenols in order of increasing acidity, and explain your answer: CH
-
Reduction of 2-butanone with NaBH4 yields 2-butanol. Is the product chiral is it optically active? Explain.
-
Plot resonance curves for the circuit of Fig. 15-29(a) for R = 2, 4, 6, 8, and 10 . I 2 120 mA ac (a) L 10 mH R C 1 F
-
An of particles is doubled and the pressure and temperatu 7:27 Draw a picture that represents the given balloon when each of the following changes occurs. volumes increase a. The balloon is inflated...
-
Required: Based on the information in the table below, prepare an income statement for Windswept Woodworks, Incorporated for year 2. Assume that the company pays a flat tax rate of 21% on its taxable...
-
This problem is adapted from a problem in CodeForces, the premier Russian competitive coding problem collection and online judge. Even if you never participate in competitive coding contests, we can...
-
In the Background section, it says that the "copper(II) concentration in the Cu(103)2(S) Cu+ (aq) + 2103(aq) equilibrium solution can be determined spectroscopically; it tells you that you will...
-
Integrative - Expected return, standard deviation, and coefficient of variation Perth Industries is studying an asset. The probability distribution of retums for this asset is shown in the following...
-
Describe how present values of uneven cash flow streams are calculated using a regular calculator and using a spreadsheet.
-
Refer to the information from Exercise 22-19. Use the information to determine the (1) Weighted average contribution margin , (2) Break-even point in units, and (3) Number of units of each product...
-
The enthalpy of formation of NaI(s) is -288 kJ mol -1 . Use this value, together with other data in the text, to calculate the lattice energy of NaI(s).
-
List the following compounds in order of decreasing reactivity toward CH3O- in an SN2 reaction carried out in CH3OH: CH3F, CH3Cl, CH3Br, CH3I, CH3OSO2CF3, 14CH3OH.
-
Starting with (S)-2-bromobutane, outline syntheses of each of the following compounds: (a) (b) (c) (d) (R)-CH3CHCH2CH3 OCH2CH3 (R)-CH3CHCH2CH3 CCH3 (R)-CH3CHCH2CH3 SH (R)-CH3CHCH2CH3 SCH3
-
Which alkyl halide would you expect to react more rapidly by an SN2 mechanism? Explain your answer. (a) (b) (c) (d) (e) Br r or or CI
-
Compute the following 6 answers for this bong paying semi-annual interest given:Par Value = $1,000Coupon Rate = 10.80%Time to Mature = 12 yearsFirst Opportunity to Call = 6 yearsCall Premium =...
-
The following are check figures: From the ledger - ending cash balance=$22,570. Using the 10 column worksheet find the remaining balances. The figures include the - Trial balance totals=$85,595,...
-
Leadership Identity Theory Instructions Researchers (i.e. Komives, Longerbeam, Mainella, Osteen, and Owen) have developed a theoretical framework to help individuals develop their leadership...

Study smarter with the SolutionInn App


