The oxygen in water is primarily (99.8%) 16O, but water enriched with the heavy isotope 18O is
Question:
The oxygen in water is primarily (99.8%) 16O, but water enriched with the heavy isotope 18O is also available. When an aldehyde or ketone is dissolved in 18O-enriched water, the isotopic label becomes incorporated into the carbonyl group.Explain.
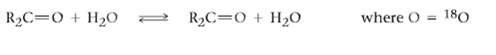
Transcribed Image Text:
R2C=0 + H2O where O %3D 180 R2C=0 + H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
0 OH ROH R This exchange present OH The above mechanism is similar to ot...View the full answer

Answered By

Arshad Ahmad
Well, I am really new to tutoring but I truly believe a good student can be a better teacher. I have always been a topper at school. I passed my Chartered Accountancy at a very young age of 23, a rare feat for most of the students. I am really dedicated to whatever work I do and I am very strict regarding deadlines. i am always committed and dedicated to whatever work allotted to me and I make sure it is completed well within deadline and also I try to give my best in whatever I do. Hope we will have a good time studying together.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The reaction of an aldehyde or ketone with a Grignard reagent is a nucleophilic addition to the carbon-oxygen double bond. (a) What is the nucleophile? (b) The magnesium portion of the Grignard...
-
When an aldehyde or a ketone is condensed with ethyl a-chloroacetate in the presence of sodium ethoxide, the product is an α,β-epoxy ester called a glycidic ester. The...
-
Treatment of an aldehyde or ketone with cyanide ion (: C N), followed by proton-ation of the tetrahedral alkoxide ion intermediate, gives a cyanohydrins. Show the structure of the cyanohydrins...
-
Why do countries respond differently in terms of flexibility and worklife balance to what seem similar economic pressures?
-
Imari Brown is attending community college. She has $1,000 of education expenses. She claims herself on her tax return. She is trying to decide between the tuition and fees deduction or an education...
-
Distinguish between common stock and retained earnings.
-
A milk processing unit claims that, of the processed milk converted to powdered milk, \(95 \%\) does not spoil. Find the probabilities that among 15 samples of powdered milk (a) all 15 will not...
-
Firms often enter into transactions that are peripheral to their core operations but generate gains and losses that must be reported on the income statement. A gain labeled peripheral by one firm may...
-
Image transcription text Problem 2 Consider the following cantilever beam in Figure P2a, which is supported by a fixed support at point O. The beam is loaded with a diagonal force F = 950 N at point...
-
In a recent years financial statements, Home Depot reported the following results. Compute and interpret Home Depots return on assets (assume competitors average a 5% return on assets). Sales . . . ....
-
When dissolved in water, trichloroacetaldehyde (chloral, CCl3CHO) exists primarily as chloral hydrate, CCl3CH (OH)2, better known as knockout drops. Show the structure of chloral hydrate.
-
Cyclohexanone forms cyanohydrins in good yield but 2, 2, 6-trimethylcyclo-hexanone does not. Explain.
-
Use the method of cylindrical shells to find the volume generated by rotating the region bounded by the given curves about the y-axis. y = 1/x, y = 0, x = 1, x = 4
-
How would the RAISE Actpromote institutional racism if made into law?
-
Please answer the following questions: 1.Define remedy as it relates to a court of law. How does the criminal law operate as a remedy? How does civil litigation act as a remedy? 2.What are the...
-
What role does historical migration and diaspora play in shaping the ethnic composition of communities and nations?
-
Explain aristotles theory of law and platos theory of law.?
-
How does ethnic identity intersect with broader social constructs such as race and nationality in contemporary societies?
-
Prove Corollary 6.2. That is, let \(\left\{\left\{X_{n k}ight\}_{k=1}^{n}ight\}_{n=1}^{\infty}\) be a triangular array where \(X_{11}, \ldots, X_{n 1}\) are mutually independent random variables for...
-
At Glass Company, materials are added at the beginning of the process and conversion costs are added uniformly. Work in process, beginning: Number of units Transferred - in costs Direct materials...
-
(A) Acetonitrile is an industrial solvent. Propose a hybridization and bonding scheme consistent with its structure. (B) A reference source on molecular structures lists the following data for...
-
(a) Provide the reagents required to accomplish the following transformation. (b) What product would you likely obtain if you attempted to synthesize the nitrile above by the following method? CO,H...
-
Write structures for the products of the following reactions: (a) C6H5CH2OH + C6H5N==C==O : (b) ClCOCl + excess CH3NH2 : (c) Glycine (H3+NCH2CO2-) + C6H5CH2OCOCI HO:- (d) Product of (c) + H2, Pd :...
-
Using decarboxylation reactions outline a synthesis of each of the following from appropriate starting materials: (a) 2-Hexanone (b) 2-Methylbutanoic acid (c) Cyclohexanone (d) Pentanoic acid
-
Calendar Plc have a year end of 30/09/X2. They have prepared the following information about their non-current assets: Land was revalued to 4.2 million on 28/09/X2 All buildings are depreciated using...
-
Antique Accents reported the following on its 2020 income statement. Using the income statement and the balance sheet given below, prepare the statement of cash flows using the indirect method. For...
-
Jamal, Maggie, and Kendra are dividing an estate consisting of a house, a vacation home, and a small business. Their valuations are shown below. Determine the payment that Kendra receives from the...

Study smarter with the SolutionInn App


