Explain which of the two following structures would be more stable. Explain whether they represent isomers or
Question:
Explain which of the two following structures would be more stable. Explain whether they represent isomers or are resonance structures.
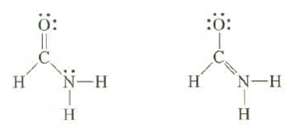
Transcribed Image Text:
HIN: H N-H H :0: N-H HIN Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
These are resonance structures because the positions of al...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the two isomers in each of the following sets should have the greater basicity at the carbonyl oxygen? Explain. H,C CHCHC- OCH or HCC-o-CH CH CH
-
Explain which of the following approaches to strategy formulation is more likely to generate economic profits: (a) Evaluating external opportunities and threats and then developing resources and...
-
Explain which of the following is a two-tailed test, a left-tailed test, or a right-tailed test. a. H0: = 12, H1: < 12 b. H0: < 85, H1: > 85 c. H0: = 33, H1: 33 Show the rejection and...
-
A list of all of the raw materials needed to manufacture a job. Question 5 options: Bill of Materials Job costing Job cost record Question 6 (1 point) A system for assigning costs to products or...
-
Explain the meaning of customer response time and its four components. Are these measures relevant to a service firm? Illustrate your answer with an example.
-
A factorial experiment was designed to test for any significant differences in the time needed to perform English to foreign language translations with two computerized language translators. Because...
-
Frequently, sales managers use contests and recognition rewards to motivate the sales force. If sales managers understand salesperson performance, why is it necessary to employ these additional...
-
Peach Corporation (a calendar year company) recorded the following transactions. Taxable income ...................... $5,000,000 Regular tax depreciation on realty in excess of ADS (placed in...
-
1 2. Let A(x) = sin t + 1 dt, find A'(x) at x = 0, and 2 3. Evaluate the following definite integrals: 2 (a) (3x + 4x)dx 4 (b) xdx
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c) H-C-N=N: H e) H H-=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
-
Draw a Lewis structure for carbon monoxide (CO). Calculate the formal charges on the atoms and comment on the stability of this compound.
-
Classify each equation as separable, linear, both, or neither. dy dx + x = xy
-
On 15 January 2009, a US Airways plane crashed into the Hudson River in New York. Janis Krums, who was on the rescue boat, tweeted the message There's a plane in the Hudson. I'm on the ferry going to...
-
An article titled Does Vasectomy Cause Prostate Cancer? (Chance, Vol. 10, No. 1) reports on several large studies that found an increased risk of prostate cancer among men with vasectomies. In the...
-
Consider the following two choices: 1. You have to choose between losing \(\$ 7400\) for sure, and a risky alternative, whereby you lose \(\$ 10,000\) with probability 0.75 and nothing with...
-
There is a strong correlation between tobacco smoking and incidence of lung cancer, and most physicians believe that tobacco smoking causes lung cancer. Yet, not everyone who smokes gets lung cancer....
-
Which of the following describes the charges on the droplets that end up in the five tubes, moving from left to right? A. \(+2 q,+q, 0,-q,-2 q\) B. \(+q,+2 q, 0,-2 q,-q\) C. \(-q,-2 q, 0,+2 q,+q\) D....
-
Ventura Ltd is considering launching a new product. It will do so if the expected value of the total revenue is in excess of $\$ 1,000$. It is decided to set the selling price at $\$ 10$. After some...
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
A function f is defined by Evaluate f(0), f(1), and f(2) and sketch the graph. f(x) 1- 1.1. x if x 1 if x > 1
-
There are two different substances with the formula C2H6O. Draw both, and tell how they differ.
-
There are two different substances that contain a carbon-carbon double bond and have the formula C4H8. Draw them, and tell how they differ.
-
Among the most common over-the-counter drugs you might find in a medicine cabinet are mild pain relievers such ibuprofen (Advil, Motrin), naproxen (Aleve), and acetaminophen (Tylenol). (a) How many...
-
Why are gains/losses added back to (or deducted from) the cash from operations section of a cash flow statement?
-
Combination A, B, C, and F is the fifth most valuable with an NPV of $169,000. how did you came up with it?
-
Using a suitable example explain how you would check that equipment is ready for shut down to commence?

Study smarter with the SolutionInn App


