Treatment of 1, 1-diphenyl- 1, 2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a
Question:
Treatment of 1, 1-diphenyl- 1, 2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction.
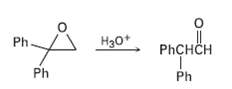
Transcribed Image Text:
Ph H30+ PHCHCH Ph Ph
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
HOH PhCCH PhCCH OH PhCCH HO PhCHCH H h...View the full answer

Answered By

Rehab Rahim
I am well versed in communicating and teaching in areas of all business subjects. I have helped many students in different ways from answering answers to writing their academic papers.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of benzoic acid with oxalyl chloride. This mechanism begins like the thionyl chloride reaction, to give a reactive mixed anhydride. Nucleophilic acyl substitution...
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Propose a mechanism for the reaction of (a) 1-methylcyclohexanol with HBr to form 1-bromo-1-methylcyclohexane. (b) 2-cyclohexylethanol with HBr to form 1-bromo-2-cyclohexylethane?
-
U Ltd has three subsidiaries (one of which is dormant) and has the following results for the year to 31 December 2020: Compute the corporation tax liability for the year and state the date (or dates)...
-
Describe the types of information employees at an Apple store require and compare it to the types of information the executives at Apple's corporate headquarters require. Are there any links between...
-
Why does the existence of a potent Pigou Effect guarantee a negatively sloped AD curve?
-
A cylindrical volume of space contains a uniform magnetic field of magnitude \(0.12 \mathrm{~T}\) but unknown direction. If the dimensions of the cylindrical volume are length \(\ell=0.060...
-
The year-end adjusted trial balance of Hilltoppers Corporation included the following account balances: Cash, $5,000; Equipment, $17,000; Accounts Payable, $3,000; Common Stock, $11,000; Retained...
-
16. 17. The IUPAC nomenclature of an element with electronic configuration [Rn] 5f46d17s is: (a) Unnibium (b) Unnilunium (c) Unnilquandium (d) Unniltrium The compound(s) that is (are) removed as slag...
-
Gloria Suarez is a 31-year-old single mother who contacted the Gulf Coast Counseling Center concerning therapy for herself. She told the intake worker that she was feeling very down and exhausted and...
-
How would you synthesize racemic disparlure (Problem 18.46) from corn- pounds having ten or fewer carbons?
-
How would you prepare o-hydroxyphenyl-acetaldehyde from phenol? More than one step is required. HO o-Hydroxyphenylacetaldehyde CH-CO
-
In June 2007, Deutsche Post World Net acquired 49 percent of the shares of U.S. airline ASTAR Air Cargo Holdings for 67 million. ASTAR is consolidated by Deutsche Post. Following is information on...
-
$8.30/hour for producing at least 550 units . $10.70/hour for producing at least 720 units $13.20/hour for producing at least 1,000 units $17.70/hour for producing at least 1,310 units All employees...
-
Implement the following program: 1) Define a class Director with two attributes name and numFilms. 2) Define an init function for Director which sets the attribute value via input parameters. 3)...
-
You have mean-variance preferences of the form U = -a02. Moreover, if you have $20, you are indifferent between a lottery that pays you $80 with probability 0.2, and $10 otherwise, and a lottery that...
-
Is the App appealing to all age groups? Lion Foods has a high proportion of older customers and the company has tried to attract them to using the App by making a lot of the Boost promotions...
-
Calculate and compare the Pepsi and Coke based on the broader ratios. BALANCE SHEET Pepsi Coke Assets 2019 2019 Cash 7,282 9,000 Short term investment 0 48,600 Acc. Receivable 632,160 351,200...
-
What is the most uncertain variable in MMCs capitated plan pricing analysis?
-
Swifty company is a publicly held corporation whose $1 par value stock is actively traded at $30 per share. The company issued 3400 shares of stock to acquire land recently advertised at $93000. When...
-
In a foundry, metal castings are cooled by quenching in an oil bath. Typically, a casting weighing 20 kg and at a temperature of 450 C is cooled by placing it in a 150-kg involatile oil bath...
-
Use arguments based on resonance and electronegativity effects to explain the trend in carbonyl IR stretching frequencies from higher frequency for esters and carboxylic acids to lower frequencies...
-
Classify each of the following compounds as an alkane, alkene, alkyne, alcohol, aldehyde, amine, and so forth. (a) (b) CH3-C¡CH (c) (d) (e) (f) Obtained from oil of cloves -2. Sex attractant...
-
Boron trifluoride (BF3) has no dipole moment ( = 0 D). Explain how this observation confirms the geometry of BF3 predicted by VSEPR theory.
-
What results have researchers found on what therapy is better clinical psychology therapists or positive psychology therapists?
-
PAIBOC Questions and analysis Scenario: Seneca has decided to hold a health promotion day for students and faculty next month. In preparation for this event, Seneca students are invited to share...
-
What is Clinical Psychology? Answers I specifically have are: What does this specialty work on specifically? How do the different types of psychologies relate to the study Clinical Psychologists do?...

Study smarter with the SolutionInn App


