Classify each of the labeled bonds in the following structure in terms of the bond type (
Question:
Classify each of the labeled bonds in the following structure in terms of the bond type (σ or π) and the component orbitals that overlap to form the bond. (For example, the carbon--carbon bond in ethane is an sp3-sp3 a bond.)
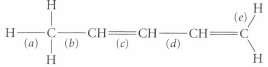
Transcribed Image Text:
(el ta) (b) lc)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
a An sp 3 C1sH s bond b An sp ...View the full answer

Answered By

Jason J Pulikkottil
I am currently pursuing a Master's degree in Computer Applications.I have completed a professional diploma in IT infrastructure management.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Classify each of the following statements as true or false: (a) For molecular solids the melting point generally increases as the strengths of the covalent bonds increase. (b) For molecular solids...
-
Classify each of the following as a primary, secondary, or tertiary alcohol. a. b. c. d. CH3 CH3 CH3CCH2CHOH CH CH,OH CH,CH2CCH CH3 CH2CH3 CH3CH2COH CH2CH3
-
Classify each of the following statements as true or false: (a) Although both molecular solids and covalent-network solids have covalent bonds, the melting points of molecular solids are much lower...
-
The manufactured wood beam carries a uniformly distributed load of in- tensity wo. Determine the largest safe value of wo if the maximum shear stress in the wood is limited to 300 psi. 1.0 ft Wo 4 ft...
-
Consider an argument you have recently had with a friend, family member, manager, co-worker, or someone else. Identify the topic of the argument and present that argument in premise-conclusion form,...
-
A Jones has the following credit purchases and credit sales for May: Required: (a) Write up sales and purchases day books. (b) Post the items to the personal accounts. (c) Post the totals of the day...
-
To what degrees are endogamy and exogamy practiced within American society? What kinds of groups are endogamous and exogamous?
-
Bond A pays $8,000 in 20 years. Bond B pays $8,000 in 40 years. (To keep things simple, assume these are zero-coupon bonds, which means the $8,000 is the only payment the bond holder receives.) a. If...
-
Write an algorithm/pseudo code to transpose an nxn matrix.
-
The file Problem5_9.xlsx contains passing statistics for NFL teams during the 2017 season. Using formulas that do combine INDEX and MATCH in the same cell, return the desired statistics in the range...
-
Classify the compounds within each of the following pairs as either identical molecules (I), constitutional isomers (C), stereoisomers (S), or none of the above (N). (a) cyclopentane and cyclopentene...
-
Give the structure of: 2E,7Z)-5-[(4- 1-propenyl]-2,7 -non adiene Be sure to read Study Guide Link 4.2 if you have difficulty with this problem.
-
On the fifth floor of the physics building, you are in a laboratory class studying induction. You are using a computer to measure emf values from a solenoid when suddenly a thunderstorm breaks out....
-
What are Robert Gagne's Learning Outcome? Describe adult learning theory. Describe discussion method. What do the trainer do in discussion method. What are the training evaluation variables and how...
-
State how many complex and real zeros the function has. f(x) = x + 8x- 8x +96x-240 The number of complex zeros are The number of real zeros are
-
TSLA is trading at $ 2000.00. In 1 year it can either go up to $ 3000 or down to $ 1500. If the current 1-year t-bill is yielding 0.35%, find the price of a 1year call option with an exercise price...
-
ABC Company is considering the purchase of a new machine for $64,000 installed. The machine will be depreciated by MACRS as 5 year property. The firm expects to operate the machine for 4 years and...
-
A treasury note with a coupon rate of 16 1/4% that settles on February 15th, 2022 and matures on August 15, 2023. Assuming a quoted price on the note is 104.25, what is the yield on the note (using...
-
A heavy object of weight W is dropped onto the midpoint of a simple beam AB from a height h. Obtain a formula for the maximum bending stress s max due to the falling weight in terms of h, st , and ...
-
Subprime loans have higher loss rates than many other types of loans. Explain why lenders offer subprime loans. Describe the characteristics of the typical borrower in a subprime consumer loan.
-
Which of the following orbitals is (are) not permitted by the quantum theory of the hydrogen atom? Explain. 2s 6s 5d 2d 3p
-
Give the electronic configuration of (a) The chlorine atom; (b) The silicon atom (Si); (c) The argon atom; (d) The magnesium atom. Indicate the valence electrons and the valence orbitals of Si.
-
Predict the approximate bond angles in each of the following molecules, and explain your reasoning. (a) :CH (b) BeH (c) *CH3 (d):CLSi (e) -0-0 (f) HC=C=CH allene HC-N OF ozone :O: (Give H-C-C and...
-
Check that the angle in your calculator is in "degrees", sin30 = 0.5 Part A - The magnitude of the normal force FN has been calculated ro be FN = 9.70 N. Calculate the work done by the normal force....
-
The eye of a hurricane passes over Grand Bahama Island in a direction 60.0 north of west with a speed of 42.0 km/h. Three hours later, the course of the hurricane suddenly shifts due north, and its...
-
Three vectors are shown in the figure below, but they are not drawn to scale. The sum of the three vectors is = F + 2 + 3. If Ry = 0 and F2 = 0.6F3, find Rx in terms of F. Rx F1 F2 30% F F.

Study smarter with the SolutionInn App


