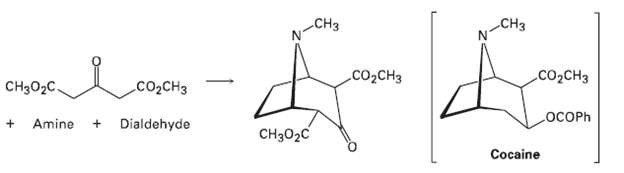
Cocaine has been prepared by a sequence beginning with a Mannich reaction between dimethyl acetonedicarboxylate, an amine,
Question:
Cocaine has been prepared by a sequence beginning with a Mannich reaction between dimethyl acetonedicarboxylate, an amine, and a di-aldehyde. Show the structures of the amine anddi-aldehyde.
Transcribed Image Text:
СНз CHз alo CO2CH3 CO2CH3 Cо,снз CHз02C. LOCOPH Dialdehyde Amine Cнз02C Cocaine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
The Mannich reaction occurs ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following profit reconciliation statement has been prepared by the management accountant of ABC Limited for March: The standard cost card for the company's only product is as follows: ()...
-
The following balance sheet has been prepared by the accountant for Limestone Company as of June 3, 2013, the date on which the company is to file a voluntary petition of bankruptcy: LIMESTONE...
-
The following balance sheet has been prepared by the accountant for Limestone Company as of June 3, 2017, the date on which the company is to file a voluntary petition of bankruptcy: LIMESTONE...
-
Suppose that the vice president of marketing asks you to write a program to create labels for a onetime advertising promotion. As IT manager, you know that the labels can be prepared more efficiently...
-
What strategies do college students employ in order to "game" their academic report cards? In light of this gaming, how can prospective employers and graduate schools determine the true academic...
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
What is the difference between the goals of a business-strategy and an IT strategy?
-
Douglas Manufacturing Company has two production departments: Cutting and Assembly. July 1 inventories are Raw Materials $4,200, Work in Process-Cutting $2,900, Work in Process-Assembly $10,600, and...
-
Victoria is developing symptoms of senile dementia and there are times when she does not fully grasp the consequences nor the reality of her actions Victoria hears noise in attic and calls Sam'...
-
1. Advise Sara on whether she is really learning some valuable lessons as a potential manager. 2. What should Sara tell Johnny on her review of her experiences? 3. Which managerial roles has Sara...
-
The Mannich reaction of a ketone, an amine, and an aldehyde is one of the few three-component reactions in organic chemistry. Cyclohexanone, for example, reacts with dim-ethylamine and acetaldehyde...
-
The male sex hormone testosterone contains C, H, and O and has a mass of 288.2089 amu as determined by high-resolution mass spectrometry. What is the likely molecular formula of testosterone?
-
What is the wavelength of a photon that has the same momentum as an electron moving with a speed of 1500 m/s?
-
What are the key principles of microservices architecture, and how does it differ from a monolithic architecture? Discuss the challenges of maintaining data consistency and service communication in a...
-
Suppose that a new trade agreement between Australia and Canada causes the exchange rate to change from CAD 0.9 /AUD to AUD1.25/CAD. What has happened to the AUD relative to the CAD? It has...
-
(i) Is the Marxist analysis valid to understand the divergences in development styles and their relationship with the international market? (ii) How are the production of surplus value and the...
-
Suppose that the wheat market is perfectly competitive. Market demand for wheat is given by the expression. = 80 5, where P is the market price per bag of wheat. The short-run market supply for wheat...
-
In a small economy, the demand and supply of organic olive oil are given by the following functions: Qd=120 000-1000P and P=20+Qs/9000, where P is the price per liter, in euros. The price of olive...
-
Big Ben in London is the most accurate mechanical clock of its size. The \(300-\mathrm{kg}\) hour hand is \(2.7 \mathrm{~m}\) long, and the \(100-\mathrm{kg}\) minute hand is \(4.2 \mathrm{~m}\)...
-
Air pollution generated by a steel mill is an example of a) a positive production externality. b) a negative production externality. c) a public good. d) the free-rider problem. State and local taxes...
-
Write a Lewis structure for the NO 3 ion. Include resonance structures.
-
Show how you would synthesize each compound, starting with alkenes or cycloalkenes that contain no more than six carbon atoms. You may use any additional reagents you need. (a) (b) (c)
-
Using 1,2-dimethylcyclohexene as your starting material, show how you would synthesize the following compounds. (Once you have shown how to synthesize a compound, you may use it as the starting...
-
Predict the major products of the following reactions, and give the structures of any intermediates. Include stereochemistry where appropriate. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n)...
-
What factors contribute to the formation of high-performance teams, and how can organizations cultivate and sustain these dynamics over time ?
-
The standard 2-pass spur gear train with the # of teeth on each gear as shown is driven is driven at 1000 RPM from shaft 2 with a torque of 25 in-lb. All the gears in the set have a diametral pitch...
-
Marigold, Inc. reports all its sales on credit, and pays operating costs in the month incurred. Amounts for 2022 are: March April May June July Budgeted sales $299500 $290000 $319700 $279100 $209800...

Study smarter with the SolutionInn App


