Complete the following reactions. OH NO2 Cr(VI) HSO4
Question:
Complete the following reactions.
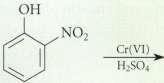
Transcribed Image Text:
OH NO2 Cr(VI) HSO4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)
A paraquinon...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Complete the following nuclear reactions: (c) 1 (o, p)
-
Complete the following nuclear reactions and find their Q values (use Appendix V for masses if necessary): (a) Li +_ (b) U + in (c) Be(a, %C He + He 5(dn) + Sr
-
Complete the following nuclear reactions. a. b. c. ISP + ? Si 14
-
Suppose the 2017 adidas financial statements contain the following selected data (in millions). Compute the following values and provide a brief interpretation of each. (a) Working capital. (b)...
-
Delta Company has 100,000 shares outstanding and plans to pay $1.00 per share in dividends each quarter next year. Delta has a capital budget of $700,000 for next year and plans to maintain its...
-
The prime entry for the acquisition of a Cash Book, ledgers and a journal for 240 from W. Smith Ltd on credit would be in the: (a) Purchases Day Book (b) Journal (c) Cash Book (d) None of the above
-
The 50-lb weight is falling \(3 \mathrm{ft} / \mathrm{s}\) at the instant it is \(2 \mathrm{ft}\) above the spring and post assembly. Determine the maximum stress in the post if the spring has a...
-
Audrinas Fleet Feet, Inc., produces dance shoes for stores all over the world. While the pairs of shoes are boxed individually, they are crated and shipped in batches. The shipping department records...
-
Cullumber guarantees a residual value of $10,000 at the end of the lease term. What if the expected residual value at the end of the lease term is $7,000 and Cullumber guarantees a residual of...
-
On January 1, 2020, Innovus, Inc., acquired 100 percent of the common stock of ChipTech Company for $670,000 in cash and other fair-value consideration. ChipTechs fair value was allocated among its...
-
Which of the, two phenols in each set is more acidic? Explain. Phenol or m-chlorophenol
-
Draw the important resonance structures of the radicals formed when each of the following react with R, a general free radical. BHT
-
Imagine that you have decided to run a direct business online. What features would you want to include in your website to attract as many customers as possible, and maximise your revenue? Before you...
-
A company produces shirts and sells them on average for $40 with a unit contribution margin of 32%. If the total fixed costs are $300,000: What is the breakeven point? How many units must be sold to...
-
Calculate the price an American call option on a non dividend paying stock with the information below. Stock price is $40 Exercise price is $38 Risk free rate is 5% Volatility is 20% Six months to...
-
Weghorst Co. is considering a three-year project that will require an initial investment of $55,000. It has estimated that the annual cash flows for the project under good conditions will be $80,000...
-
Stay Swift Corp. is looking at investing in a production facility that will require an initial investment of $500,000. The facility will have a three-year useful life, and it will not have any...
-
Magnetis Inc. has annual sales of $12 million, its cost of goods sold is 75% of annual sales, it carries an average inventory of $800,000 and its average collection period is as long as its inventory...
-
Decide whether the limits in Exercises exist. If a limit exists, find its value. (a) (b) (c) (d) g(-1) lim_g(x) x-1
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
You need to know the melting point for CaC1 2 , for a lab report you are writing. Your lab partner says that the Handbook of Chemistry and Physics lists this as 68 o C. Do you think you should trust...
-
Show a Lewis structure for A1C1 4 . What are the formal charges on the atoms of this anion? What is its shape?
-
Ammonium cyanate is composed of an ammonium caution (NH 4 + ) and a cyanate anion (OCN ). Show a Lewis structure for the cyanate anion. (Both O and N are bonded to C.) Which atom has the negative...
-
Should an MNC purchase Nestl stock yes or no? Recommendations should include the company's sustainability plans, the image of the company, Gordon Growth result, and financial performance. Any...
-
Can the sponsor, or general partner, continue to achieve such high returns? Framed another way, can the general partner earn a 25+% return on the $6,305,000 received at the end of the 10th year? If...
-
Select a hospital that publicly displays their financial performance on their website. Describe the Profit Loss Statement: Review top revenue items Review cost comparison compared to previous year...

Study smarter with the SolutionInn App


