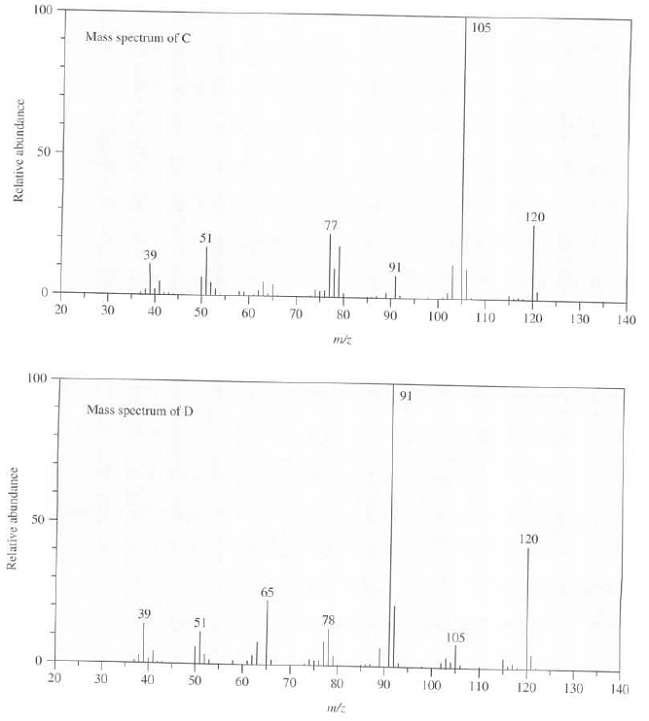
Compounds C and D are isomers with the formula C9H12, in addition to other absorption peaks, both
Question:
Compounds C and D are isomers with the formula C9H12, in addition to other absorption peaks, both compounds show a peak near 7.25 ? (area 5) in their 1H-NMR spectra. Their mass spectra are as follow, show the structures of C and D.
Transcribed Image Text:
100 105 Mass spectrum of C 50 120 77 31 39 91 20 30 40 50 70 60 120 80 90 100 110 130 140 100 91 Mass spectrum of D 50 120 65 39 78 51 105 0. 20 30 40 50 60 70 80 90 100 110 120 130 140 Relative abundance Relative abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
The isomers have DU 4 The 1 HNMR peak near 725 area 5 indicates the presence ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Compounds A, B, and C are isomers with the formula C5H11Br. Their broadband proton-decoupled 13C NMR spectra are given in Fig. 9.32. Information from the DEPT 13C NMR spectra is given near each peak....
-
Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm ?1 in its IR spectrum and B has a peak at 1715cm ?1 , the mass spectra of A and B are as follows, show the structures of...
-
Compounds Y and Z are isomers with the molecular formula C10H12O. The IR spectrum of each compound shows a strong absorption band near 1710 cm-1. The 1H NMR spectra of Y and Z are given in Figs. 16.4...
-
Write a check (Figure 4 - 42) dated June 13, 20XX, to Byron Johnson in the amount of $296.83 for a washing machine, Complete the check stub. Figure 4 - 42 456 456 Amount To For KRA, INC 2596 Jason...
-
From what you have learnt so far, briefly explain the key responsibilities of public administrators and the best way of meeting the responsibilities.
-
In ways does ESPN help develop their employees for future success?
-
Calculate the interquartile range for each of the following sets of data: a. \(3,6,7,12,15,17,23,28\) b. \(8,4,1,6,13,10,12,5\) c. \(3,8,14,11,16,7,14,15,11,9,12,6\) d....
-
During December 2010, Amin Corp. manufactured products requiring 8,000 standard labor hours. The following variance and actual information is available: Labor rate variance ........ $4,500 U Labor...
-
Pelican Paint Maufacturers, Inc. uses process costing for its mixing department. It mixed 5 , 0 0 0 units of paint during the period and has 6 0 0 are still being processed ( ending WIP ) in the...
-
Consider the following 0x86 program: .data array DWORD 1,2,3,4,5,6,7,8,9 aravSize -(S-array)/4 ; array .code main PROC mov ecx,arraySize-1 mov esi.OFFSET array L1: mov eax. [esi] sda mov bx,2 idiv bx...
-
The mass spectra of 3-ethyl-2-pentanone and 4-methyl-2-pentanone are as follows. Explain which spectrum goes with which compound, what is the structure of the ion responsible for the peak at m/z 43...
-
Compounds E and F are isomers with the formula C6H8 both react with H2 in the presence of Pt to give G (C6H12), G shows a single peak in its 13C-NMR spectrum. E has no absorption maximum above 200nm...
-
Which of the following sentences, if added here, would most effectively introduce the subject of the remainder of the essay? A. NO CHANGE B. (Do NOT begin a new paragraph) For me, C. (Begin a new...
-
Parallel would be best so when one light goes out the rest of the lights would still be on. E. What is a current? What is a voltage? What are the SI units of each?
-
2. A system consisting of four particles obeying Maxwell-Boltzmann distribution with a total energy of 48. The degeneracy of the energy levels is two. With the help of a diagram, (a) (b) (c)...
-
When we are solving thin film interference problems, we compare the wavelength of the incident light to the thickness of the film. Which value should we use for the wavelength? O The value of the...
-
How many moles of carbon dioxide, CO2, are in a 22 gram sample of the compound?
-
13 The "normal" eye has a near point of 25 cm. If a given individual's near point is 41 cm, for what problem will the eye doctor prescribe correction? O hyperopia O myopia O presbyopia O astigmatism...
-
Go to www.project-management-podcast.com/index.php/podcast-episodes/episode-details/109-episode-063-how-do-risk-attitudes-affect-your-project to access the podcast on risk attitudes on projects. What...
-
When you weigh yourself on good old terra firma (solid ground), your weight is 142 lb. In an elevator your apparent weight is 121 lb. What are the direction and magnitude of the elevator's...
-
Some policymakers in the United States have argued in favor of eliminating $100 bills in order to make large cash transactions undertaken by drug dealers more difficult. Could the former Soviet...
-
State whether you would expect the entropy change, So, to be positive, negative, or approximately zero for each of the following reactions. (Assume the reactions take place in the gas phase.) (a) A +...
-
(a) What is the value of Go for a reaction where Keq = 1? (b) Where Keq = 10? (The change in Go required to produce a 10-fold increase in the equilibrium constant is a useful term to remember.) (c)...
-
Draw contributing resonance structures and a hybrid resonance structure that explain two related facts: the carbon-oxygen bond distances in the acetate ion are the same, and the oxygen's of the...
-
Janice will need to pay $200 at the end of every month for the next 12 months, except for the payment of the 8th month. What is the present value, assuming a rate of 4%, compounded quarterly?
-
List and explain two management tools in the planning process and two measurable performance indicators. Explain in detail.
-
What does a high PE tell us about the value of the stock price (over or under valued)? What does a low PE tell us about the value of the stock price (over or under valued)? Be specific with your...

Study smarter with the SolutionInn App


