Consider the following samples of gases at the same temperature. Arrange each of these samples in order
Question:
Consider the following samples of gases at the same temperature.
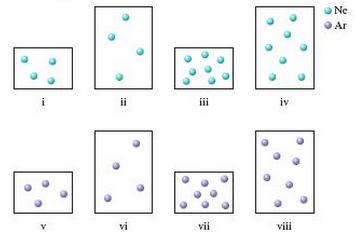
Arrange each of these samples in order from lowest to highest.
a. Pressure
b. Average kinetic energy
c. Density
d. Root mean square velocity
Some samples of gases may have equal values for these attributes. Assume the larger containers have a volume twice the volume of the smaller containers and assume the mass of an argon atom is twice the mass of a neon atom.
Transcribed Image Text:
Ne Ar iv vi vii vili
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
a Containers ii iv vi and viii have volumes twice those of containers i iii v and vii Containers iii ...View the full answer

Answered By

Simon kingori
I am a tier-one market researcher and content developer who has been in this field for the last six years. I’ve run the freelancing gamut; from market research, data mining and SEO/SMM to copywriting, Content Development, you name it, I’ve done it. I’m extremely motivated, organized and disciplined – you have to be to work from home. My experience in Freelancing is invaluable- but what makes me a cut above the rest is my passion to deliver quality results to all my clients- it’s important to note, I've never had a dissatisfied client. Backed by a Masters degree in Computer Science from MOI university, I have the required skill set and burning passion and desire to deliver the best results for my clients. This is the reason why I am a cut above the rest. Having taken a Bsc. in computer science and statistics, I deal with all round fields in the IT category. It is a field i enjoy working in as it is dynamic and new things present themselves every day for research and exploration.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physical Chemistry questions
-
For each of the following samples of paired ranks, sketch a scatter diagram, find the value of rs, and state whether there appears to be a correlation between x and y. a. b. c. x | 1 3 5 4 2 y 3 5 4...
-
Consider the following samples of gases: If the three samples are all at the same temperature, rank them with respect to (a) Total pressure, (b) Partial pressure of helium, (c) Density, (d) Average...
-
Rank the following assets from lowest to highest liquidity risk: a. Three- month Treasury bills with one- year construction loan b. Four- year car loan with monthly payments c. Five- year Treasury...
-
You are the manager of an educational facility and have beenasked to predict analyze the electric cost for 2020 based on the following 12 months of Information, You know the electric cost has both...
-
A firm uses labor and machines to produce output according to the production function f(L,M) = 4L1/2M1/2, where L is the number of units of labor used and M is the number of machines. The cost of...
-
Figure shows an engine valve driven by an overhead camshaft. The rocker arm pivots about the fixed point O and the inertia of the arm about this point is Ir. The valve mass is mv and the spring mass...
-
In 1990 Congress passed the Gun-Free School Zone Act that made it unlawful to possess a firearm in a school zone. A highschool senior carried a concealed and loaded handgun into his high school and...
-
Bolz Office Supply Company recently changed its system of internal control over cash disbursements. The system includes the following features. Instead of being unnumbered and manually prepared, all...
-
The video about sign language demonstrated: A . Most sign languages are very similar. B . All sign languages use the same gesture for the word "what". C . English - speaking countries use the same...
-
Write a program that reads a graph from a file and determines whether the graph is connected. The first line in the file contains a number that indicates the number of vertices (n). The vertices are...
-
Consider two gases, A and B, each in a 1.0- L container with both gases at the same temperature and pressure. The mass of gas A in the container is 0.34 g, and the mass of gas B in the container is...
-
Calculate the average kinetic energies of the CH 4 and N 2 molecules at 273 K and 546 K.
-
Show that for an ideal Bose gas \[\frac{1}{z}\left(\frac{\partial z}{\partial T} ight)_{P}=-\frac{5}{2 T} \frac{g_{5 / 2}(z)}{g_{3 / 2}(z)}\] compare this result with equation (7.1.36). Thence, show...
-
Eight sensors each feed eight pieces of data to a circuit which processes the data. It is concluded that as opposed to utilizing 64 sign lines, the information will be multiplexed onto eight...
-
Of all the book fines a library issued one year, 0.8% of them were for amounts greater than $10. There were 15 fines greater than $10. How many fines did the library issue that year? Enter your...
-
2. Write cardinality of each set: a. S={1, 2, 3, 4} b. S {a, b, c, d, e} C. S=0 d. S=0} TAT e. S = {0} f. S={1, 3, 900, a, Hello, 0} g. S={1, 2, 3, 4, 5, 6, ...}
-
Examples of Servant Leadership, Transformational Leadership, Transactional Leadership, Distributive Leadership Identify the four dimensions of leadership and provide an example of each area.
-
1- Apply relevant organizational behavior theories and concepts to improve work performance and job commitment. Choose at least two theories or concepts, such as expectancy theory, equity theory,...
-
Using the Fresnel equations, determine Brewsters angle, the angle of incidence at which no p-polarized light can be reflected (r 0). Before polaroid plastic polarizers were invented, polarized...
-
(a) What is the focal length of a magnifying glass that gives an angular magnification of 8.0 when the image is at infinity? (b) How far must the object be from the lens?
-
The following compound is optically inactive. Explain why.
-
Identify the reagent you would use to accomplish each of the following transformations: (a) Cyclobutanol bromocyclobutane (b) tert-Butanol tert-butyl chloride (c) Ethyl chloride ethanol
-
How many different alkenes will produce 2, 4-dimethylpentane upon hydrogenation? Draw them.
-
Large-scale industrial production - accompanied by massive technological change, expanding international communication networks, and pro-growth government policies-generated rapid economic...
-
How can this relate to individuals who have had a TBI and are currently homeless or marginally housed?
-
1. According to Marxian theory, are crises an economic process inherent to the accumulation dynamics of current capitalism? Justify your answer. 2. According to Marxian theory, what do you consider...

Study smarter with the SolutionInn App


