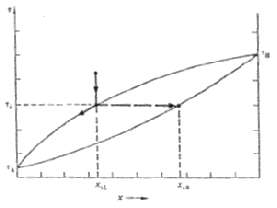
Consider the solidification of a binary alloy with the phase diagram of Figure. Show that, regardless of
Question:
Consider the solidification of a binary alloy with the phase diagram of Figure. Show that, regardless of the initial composition, the melt will always become fully depleted in component B by the time the last remnant of the melt solidifies. That is, the solidification will not be complete until the temperature has dropped toTA.
Transcribed Image Text:
i 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
Qualitatively the claimed result is all but obvious when the last infinitesimal remnant of liquid solidifies the solid forming from this remnant must ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Solid State questions
-
A thin plate of a binary alloy of composition N A = 0.245 and N B = 0.755 was welded to a similar plate of composition N A = 0.255 and N B = 0.745 so as to form a diffusion couple. After a diffusion...
-
A 5-cm-diameter sphere solidifies in 1050 s. Calculate the solidification time for a 0.3 cm ( 10 cm ( 20 cm plate cast under the same conditions. Assume that n = 2?
-
The binary phase diagram for the silver (Ag) and germanium (Ge) system is shown in Figure 11-34. Figure 11-34 The silver-germanium phase diagram (for Problem 11-35). (a) Schematically draw the phase...
-
Use the following to answer questions 1-3 below The records of Teresa's Toys Co. as of December 31, 20XX. Accounts payable Insurance expense Accounts receivable s10,000 500 19,000 S60,000 12,000...
-
You are looking at a one-year loan of $10,000. The interest rate is quoted as 8 percent plus three points. A point on a loan is simply 1 percent (one percentage point) of the loan amount. Quotes...
-
How will your answer in question #3 change if we now relax the M&M perfect capital markets assumption and incorporate a corporate tax rate of 35 percent?
-
Under Statement on Auditing Standards No. 61, "Communication with Audit Committees," which of the following would an auditor typically communicate to the audit committee? a. Difficulties encountered...
-
Cool Logos buys logo- imprinted merchandise and then sells it to university bookstores. Sales are expected to be $ 2,005,000 in September, $ 2,250,000 in October, $ 2,381,000 in November, and $...
-
A famous study on vision, conducted in 1933, presented a phenomenon known as the "Stiles-Crawford effect of the first kind" which says that the percentage P of the total luminance entering a pupil of...
-
FeVer Beverages is a typical coffee shop that operates at Batangas. Recently, the general manager is facing a challenge about not meeting their target lead time of serving the hot coffee of 15...
-
Let B be an impurity in A, with X < < 1. In this limit the non-mixing parts of the free energy can be expressed as linear functions of x, as f0(x) = f0(0) + xf0(0), for both liquid and solid phase....
-
(a) Suppose a 1000 A layer of Au is evaporated onto a Si crystal, and subsequently heated to 400oC. From the Au-Si phase diagram, Figure, estimate how deep the gold will penetrate into the silicon...
-
1. The burden of proof in a civil trial is to prove a case _____________________. The burden of proof rests with the ____________________. (a) Beyond a reasonable doubt; plaintiff (b) By a...
-
Are White male athletes characterized differently from Black athletes and/or other athletes of color? Why or why not?
-
What kinds of examples of cause-washing do you see with MLS NEXT? What about in other brands or organizations that you support?
-
Ask someone to follow a set of instructions or to fill out a form. As an alternative, you also might test a document youve created for a course. You also may try ordering food from a website, such as...
-
A rectangular loop of length \(\ell=80 \mathrm{~mm}\), width \(w=\) \(60 \mathrm{~mm}\), and resistance \(R=20 \mathrm{~V} / \mathrm{A}\) is located in a uniform magnetic field of magnitude \(B=0.50...
-
Consider a company or organization in the news that is currently responding to a crisis. Which of the five strategies of IRT (Denial, Evasion of Responsibility, Reducing Offensive, Corrective Action,...
-
log a (x 3 y) Write each expression as a sum of logs. Express powers as factors.
-
In the current year, the City of Omaha donates land worth $500,000 to Ace Corporation to induce it to locate in Omaha and create an estimated 2,000 jobs for its citizens. a. How much income, if any,...
-
Write the SQL code for the following: Update all class prices to reflect a 1% increase. a. UPDATE Reservations SET ClassPrice = ClassPrice*1.10; b. UPDATE Reservations SET ClassPrice = ClassPrice*1%;...
-
Water in an electric teakettle is boiling. The power absorbed by the water is 1.00 kW. Assuming that the pressure of vapor in the kettle equals atmospheric pressure, determine the speed of effusion...
-
A cooking vessel on a slow burner contains 10.0 kg of water and an unknown mass of ice in equilibrium at 0C at time t = 0. The temperature of the mixture is measured at various times, and the result...
-
(a) In air at 0C, a 1.60-kg copper block at 0C is set sliding at 2.50 m/s over a sheet of ice at 0C. Friction brings the block to rest. Find the mass of the ice that melts. To describe the process of...
-
The imposition of a ceiling on the length of time a criminal case should take to get to trial is a multifaceted issue, balancing the defendant's right to a timely trial with the complexities of...
-
Discuss three changes written into the Constitution in 1789 that changed the government from what it was under the ArticIes of Confederation?
-
Assume South Park is initially closed to trade. Domestic supply and demand for winter hats is given by the following equations: D = 60 - 4P, S = 2P - 6. a. Suppose South Park opens up to trade and...

Study smarter with the SolutionInn App


