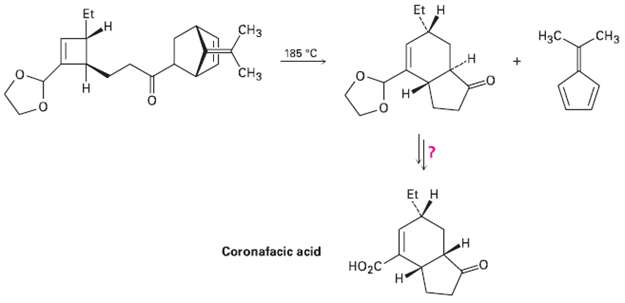
Coronafacic acid, a bacterial toxin, was synthesized using a key step that involves three sequential pericyclic reactions.
Question:
Coronafacic acid, a bacterial toxin, was synthesized using a key step that involves three sequential pericyclic reactions. Identify them, and propose a mechanism for the overall transformation. I low would you complete theSynthesis?
Transcribed Image Text:
Et H Et CHз Нас. CHз 185 "C CHз Н Et H Coronafacic acid НO2с Н и.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
Ayy Reaction 1 Reaction 2 Reaction 3 CHCH3 CHCH3 H 3 CHCH3 HC CHCH3 Reverse DielsAlder 42 ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the acid-catalyzed condensation of n-propyl alcohol to n-propyl ether, as shown above. When the temperature is allowed to rise too high, propene is formed. Propose a mechanism...
-
Propose a mechanism for the base-promoted hydrolysis of g-butyrolactone:
-
Propose a mechanism for the following reaction: + HC-o trace H CH5
-
1. You are required to compare the results of your printed reports and the results of reports of the MYOB assignment book in your MYOB assignment. If it is assumed that two results of the reports are...
-
I. Organizations should stay out of our personal lives. If we want to overeat or smoke, that is our choice. Organizations should not be allowed to monitor weight and smoking behavior. 2....
-
When are potential voting rights considered when deciding if one entity controls another?
-
Explain with neat sketches, the working of two-stroke diesel engine.
-
Ryan Patrick operates a roller skating center. He has just received the monthly bank statement at June 30 from Citizens National Bank, and the statement shows an ending balance of $750. Listed on the...
-
A bank starts with ALL of $2.965 million at the beginning of the year, charges off worthless loans of $0.436 million during the year, recovers $0.068 million on loans previously charged off, and...
-
A Queue Abstract Data type (ADT) has these associated operations: Create queue Add item to queue Remove item from queue The queue ADT is to be implemented as a linked list of nodes. Each node...
-
The sex hormone estrone has been synthesized by a route that involves the following step. Identify the pericyclic reactions involved, and propose amechanism. CH CH Heat |CH30 CH0 Estrone methyl ether
-
The following rearrangement of N-allyl-N, N-dimethyl anilinium ion has been observed. Propose amechanism. H H NH(CH3)2 eat N-Allyl-N,N-dimethylanilinium ion o-Allyl-N,N-dimethylanilinium ion
-
Assign the configuration of these compounds as R or S: a) c) e) CH3 HC C HOCH H HC g) H- B CH CH3 i) HN- -OH CH H CH3 COH FH H CH,Ph Phenylalanine b) d) f) Br CH3 "CO,H h) H- me H to Br OH -CH3...
-
Addison works for a company and pays for half of her premiums on the insurance that covers employees who need to have sick pay. This comes from after-tax dollars. She ends up getting sick for a month...
-
Entity A has the following information accounts receivable, 100,000 accounts receivable - Mr. George U. Tang, 20,000 Where will you find the accounts described above?
-
The Blum-Blum-Shub generator produces pseudorandom bits as follows. We set a large natural M, set a natural seed ro, and define +1 for each i as r(modM). We let each pseudorandom bit bi be the...
-
A taxpayer with two rental houses accounts for and/or spends the following during the year: $8,000 in depreciation deductions $7,000 in mortgage interest on the properties $3,500 in real estate taxes...
-
Post: Success in the educational setting relies heavily on the efforts of administrators, whose responsibilities may range widely depending on the nature of the institution they serve....
-
Which do you think has less variation: the IQ scores of students in your statistics class or the IQ scores of a simple random sample taken from the general population? Why?
-
Select a mass spectrometric technique with the highest mass resolution for identifying an unknown compound being eluted from a liquid chromatography column
-
Pegged orders are created to follow the NBBO as it moves. In addition to the installation of the speed bump, the IEX has provided the primary peg order, which is intended to protect small or retail...
-
Use frontier orbital analysis to decide whether the dimerization of 1,3-butadiene shown here is allowed or forbidden. 2H2C-CH-CH-CH2- s heat
-
Write structural formulas for each of the following: (a) 3,4-Octadiene (b) (E,E)-3,5-Octadiene (c) (Z,Z)-1,3-Cyclooctadiene (d) (Z,Z)-1,4-Cyclooctadiene (e) (E,E)-1,5-Cyclooctadiene (f)...
-
Give the IUPAC names for each of the following compounds: (a) CH2CH(CH2)5CHCH2 (b) (c) (CH2CH)3CH (d) (e) (f) CH2CCHCHCHCH3 (g) (h) CH3 CHz CH3 HH CI CI H H H3C CH,CH2 CH2CH3
-
A project requires an initial investment of $100,000 to purchase equipment. The equipment will be depreciated on a straight-line basis over the five-year life of the project. The company expects the...
-
Vodaphone's earnings per share in 2022 was $1.80, and in 2017 it was $1.25. The company distributes 60% of its earnings as dividends, and the stock is currently valued at $37.75. The cost to issue...
-
A bond is currently selling in the market for $1,085.96. It has a coupon of 8% and a 15-year maturity. Using annual compounding, what is the yield to maturity on this bond?

Study smarter with the SolutionInn App


