Cyclooctatetraene readily reacts with potassium metal to form the stable Cyclooctatetraene dianion, C 8 H 8 2
Question:
Cyclooctatetraene readily reacts with potassium metal to form the stable Cyclooctatetraene dianion, C8H82?1. Why do you suppose this reaction occurs so easily? What geometry do you expect for the Cyclooctatetraene dianion?
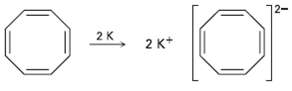
Transcribed Image Text:
2K 2 K* 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
When Cyclooctatetraene accepts two ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Why do you suppose that forensic DNA analysis relies principally on short tandem repeats (repeat polymorphisms), rather than single- nucleotide polymorphisms, such as that described on page 669 and...
-
Why do you suppose that large, well-known companies such as Apple, Starbucks, and Facebook prefer to have their shares traded on the NASDAQ rather than on one of the major listed exchanges, such as...
-
Why do you suppose that m x n machine-scheduling algorithms are not widely used in practice? Should optimal rules be more widely used?
-
Fewer than 20% of M&M candies are green. The hypothesis test results in a P-value of 0.0721. Assume a significance level of = 0.05 a. State a conclusion about the null hypothesis. b. Without using...
-
Who is affected by a hospital's decision not to hire smokers? Discuss whether this decision achieves the greatest good for the greatest number of individuals.
-
There are two dicarboxylic acids with the general formula HO2CCH==CHCO2H. One dicarboxylic acid is called maleic acid; the other is called fumaric acid. When treated with OsO4, followed by...
-
How does an auditor test whether recorded receivables represent bona fide rights of the client?
-
Agnew Chemical Company makes three products, B7, K6, and X9, which are joint products from the same materials. In a standard batch of 150,000 pounds of raw materials, the company generates 35,000...
-
A company that offers a replacement warranty had a $10,000 balance in the Warranty Liability account at year end BEFORE recording warranty expense for the year. If the balance AFTER recording the...
-
Post - Combination Balance Sheet Presented below are the balance sheets of Allen Corporation and Benson Corporation, immediately prior to a business combination. The fair values of Benson s reported...
-
Draw the five resonance structures of the cyclopentadienyl anion. Are all carbon carbon bonds equivalent? How many absorption lines would you expect to see in the 1H NMR and 13C NMR spectra of the...
-
Draw an orbital picture of furan to show how the molecule isaromatic. Furan :O:
-
How does the concept of a risk premium in incentive compensation relate to the concept of a compensating differential in compensation policy?
-
Redfern Audio produces audio equipment including headphones. At the Campus Facility, it produces two wireless models, Standard and Enhanced, which differ both in the materials and components used and...
-
Opticians use the following thin-lens formula to perform calculations related to prescription lenses. = Use this formula to determine i when f= 18 cm and o= 24 cm. Round to the nearest cm.
-
Marian Manufacturing (2M) applies manufacturing overhead to jobs based on direct labor costs. For Year 2, 2M estimates its manufacturing overhead to be $421,200 and its direct labor costs to be...
-
Clonex Labs, Incorporated, uses the weighted-average method of process costing. The following data are available for one department for October: Percent Completed Work in process, October 1 Work in...
-
Assume a parent company acquired a subsidiary on January 1, 2XX1. The purchase price was $440,000 in excess of the subsidiary's book value of Stockholders' Equity on the acquisition date, and that...
-
The proton collision that creates the pion also creates a gamma-ray photon traveling in the same direction as the pion. The photon will get to the medical bay first because it is moving faster. What...
-
Use this circle graph to answer following Exercises. 1. What fraction of areas maintained by the National Park Service are designated as National Recreation Areas? 2. What fraction of areas...
-
For the reaction, A(g) 2 B( g), K c = 4.0. A reaction mixture at equilibrium contains [A] = 1.0 M. What is the concentration of B in the reaction mixture? (a) 0.50 M (b) 1.0 M (c) 2.0 M (d) 4.0 M
-
(a) Show that D-glucose, D-mannose, and D-fructose all give the same osazone. Show the structure and stereochemistry of this osazone. (b) D-Talose is an aldohexose that gives the same osazone as...
-
Show that Ruff degradation of D-mannose gives the same aldopentose (D-arabinose) as does D-glucose.
-
D-Lyxose is formed by Ruff degradation of galactose. Give the structure of D-lyxose. Ruff degradation of D-lyxose gives D-threose. Give the structure of D-threose.
-
3. Show that : -3t = y 3 sin(t) cos(t) + e is a solution to the differential equation y' + 3 y 10 sin(t)
-
The United States, Austrailia, Canada, and the United Kingdom cluster high on individualism but low on power distance on Hofstede\'s cutlutral graph. What does this mean to someone looking to market...
-
Mini-garden project Week one of the mini-garden project involved Sacha and her fellow pre-schoolers planting sunflower seeds into the outdoor garden bed. Each child in the activity has been assigned...

Study smarter with the SolutionInn App


