Data below come from the graph in Box 14-2, for which the separate solutions method was used
Question:
Data below come from the graph in Box 14-2, for which the separate solutions method was used to measure selectivity coefficients for a sodium ion-selective electrode at 21.5°C. Use Equation 14-11 to calculate log KPot for each line below.
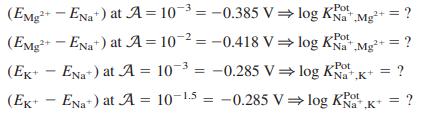
Equation 14-11
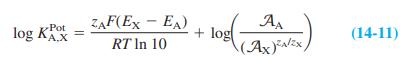
Transcribed Image Text:
(EMg+ - ENa) at A= 103 = -0.385 V = log Kot. „Mg 2+ = ? Na" Mg (EMg+ - ENa") at A= 10 = -0.418 V = log K Mg+ = ? Pot Pot (Ex+ - ENa) at A = 10 = -0.285 V = log KK = (EK+ - ENa) at A = 10-1.5 = -0.285 V log Kt Pot ,K* = ? %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
For the first line of data with A Nat and X Mg The fi...View the full answer

Answered By

ANSHUL GUPTA
I Love to interact with students in a friendly way so that student does not hesitate to ask questions to clear all his doubts. The best way to learn is that we should enjoy while studying. The most important aspect in my teaching is revision.
Presently I am working as a Credit manager in a bank. I am working in a Finance field from the past 4 years. I have gained a lot of practical knowledge in the field of Finance and Taxation and would like to share that knowledge with all of you.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Various amounts of activated carbon were added to a fixed amount of raw cane sugar solution (48 wt% sucrose in water) at 80?C. A colorimeter was used to measure the color of the solutions. R, which...
-
The following wet-ashing procedure was used to measure arsenic in organic soil samples byatomic absorption spectroscopy: A 0.1- to 0.5-g sample was heated in a 150-mL Teflon bomb in a microwave oven...
-
1. G. Johnson and colleagues have analyzed the MAP kinase cascade in which MEKK2 participates in mammalian cells. By a yeast two-hybrid screen (see Chapter 7), MEKK2 was found to bind MEK5, which can...
-
Suppose that \(\mathrm{x}\) is a linked-list Node. What is the effect of the following code fragment? \(t\). next \(=x\). next; \(x \cdot\) next \(=t ;\)
-
Suppose Kroger Co.s 2014 financial statements contained the following data (in millions). Current assets ........ $ 7,450 Total assets ........ 23,093 Current liabilities ....... 7,714 Total...
-
Extend the analysis of the laminar-flow reactor for a power-law fluid. Perform some computations using the PDEPE solver, and show how the power-law index affects the conversion in the reactor.
-
A simply supported, specially orthotropic plate is subjected to an in-plane compressive load per unit length \(N_{x}\) and an in-plane tensile load per unit length \(N_{y}=-0.5 N_{x}\) as shown in...
-
Carson Trucking is considering whether to expand its regional service center in Moab, Utah. The expansion requires the expenditure of $10,000,000 on new service equipment and would generate annual...
-
Henrich is a single taxpayer. In 2023, his taxable income is $535,500. What are his income tax and net investment income tax liability in each of the following alternative scenarios? Use Tax Rate...
-
During the month of June, Bon Voyage Travel recorded the following transactions: 1. Owners invested $25,000 in cash to start the business. They received common stock. 2. The month's rent of $500 was...
-
Ammonia in seawater was measured with an ammonia-selective electrode. A 100.0 mL aliquot of seawater was treated with 1.00 mL of 10 M NaOH to convert NH + 4 to NH 3 . Therefore,V 0 = 101.0 mL. A...
-
The H + ion-selective electrode on the Phoenix Mars Lander has selectivity coefficients K pot HNA+ = 10 -8.6 and K pot H+,Ca2 + = 10 -7.8 . Let A be the primary ion sensed by the electrode and let...
-
On the basis of the following data, estimate the cost of the merchandise inventory at April 30 by the retailmethod: Cost Retail Merchandise inventory April 1 April 1-30 April 1-30 Purchases (net)...
-
In the 2006 Winter Olympics, the men's 15-km cross-country skiing race was won by Andrus Veerpalu of Estonia in a time of 38 minutes, 1.3 seconds. Part A Determine his average speed (the distance...
-
3. Choose the correct equation for the ellipse with vertices (1, -7) and (1,3) and focus (1, 1) A) B) C) D) (x - 1) 16 (x -1) 25 (x - 3) 9 (x - 3) + + + + (y + 2) 25 (y-2) 16 (y + 2) 16 (y - 1) 25 =...
-
3. Planet 1 and 2 are in coplanar circular orbits about the sun, with average distances a and a2 respectively where a2 > a. a) Give an expression for the period of revolution about the sun for each...
-
A 11 kg beam of length 8 m holding a 19 kg mass is attached by a wire to a wall as shown. The distance of mass m from the right end of the beam is 4. The rope makes an angle of 22 degrees form the...
-
Emergency rooms in hospitals often have to treat severe cases of skin burn. Compare the severity of the burn inflicted on a person's skin by steam by comparing the heat involved in converting 15 g of...
-
When you predict that a result will be in one or another direction (for example, that a particular treatment will reduce recidivism), you would use a a. One-tailed test. b. Two-tailed test. c....
-
Separate variables and use partial fractions to solve the initial value problems in Problems 18. Use either the exact solution or a computer-generated slope field to sketch the graphs of several...
-
Pure acetic acid, known as glacial acetic acid, is a liquid with a density of 1.049 g/mL at 25C. Calculate the molarity of a solution of acetic acid made by dissolving 20.00 mL of glacial acetic acid...
-
Glycerol, C3H8O3, is a substance used extensively in the manufacture of cosmetics, foodstuffs, antifreeze, and plastics. Glycerol is a water-soluble liquid with a density of 1.2656 g/mL at 15C....
-
What mass of KCl is needed to precipitate the silver ions from 15.0 mL of 0.200 MAgNO3 solution?
-
Q5 Financial maths Initial investment: E 25,000 Year 1 return: 8,000 Year 2 return: E 15,000 Year 3 return: E Year 4 return: E 10,000 5,000 (i) Enter appropriate formulae in the shaded cells in the...
-
The production manager of Rordan Corporation has submitted the following quarterly production forecast for the upcoming fiscal year: Units to be produced 1st Quarter 10,000 2nd Quarter 7,500 3rd...
-
Annswer the following questions: [10 marks] A. Define two of the main advantages of explicit architecture. B. Compare between (1) Model-View-Controller (MVC), (1) Layered architecture pattern. The...

Study smarter with the SolutionInn App


