Demonstrate that (a) The value of ? in Equation 17.19 is 96,500 C/mol, and (b) At 25C
Question:
Demonstrate that
(a) The value of ? in Equation 17.19 is 96,500 C/mol, and
(b) At 25°C (298 K),
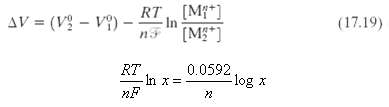
Transcribed Image Text:
RT [Mf*] (M**] AV = (V - V)- (17.19) In n3 0.0592. -log x RT -In x= nF
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a The Faraday constant represented here as F is j...View the full answer

Answered By

Anurag Agrawal
I am a highly enthusiastic person who likes to explain concepts in simplified language. Be it in my job role as a manager of 4 people or when I used to take classes for specially able kids at our university. I did this continuously for 3 years and my god, that was so fulfilling. Sometimes I've skipped my own classes just to teach these kids and help them get their fair share of opportunities, which they would have missed out on. This was the key driver for me during that time. But since I've joined my job I wasn't able to make time for my passion of teaching due to hectic schedules. But now I've made a commitment to teach for at least an hour a day.
I am highly proficient in school level math and science and reasonably good for college level. In addition to this I am especially interested in courses related to finance and economics. In quest to learn I recently gave the CFA level 1 in Dec 19, hopefully I'll clear it. Finger's crossed :)
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
A heat engine operating with an environment at 298 K produces 5 kW of power output with a first law efficiency of 50%. It has a second law efficiency of 80% and TL = 310 K. Find all the energy and...
-
At 298 K a cell reaction has a standard cell potential of +0.17 V. The equilibrium constant for the reaction is 5.5 105. What is the value of n for the reaction?
-
Demonstrate that the constant K in Equation 17.23 will have values of 534 and 87.6 for the CPR in units of mpy and mm/yr, respectively.
-
A 12-year bond has an annual coupon rate of 9%. The par value of the bond is $1,000 and the bond has a yield to maturity of 7%. Which of the following statements is correct? If the market interest...
-
John Smith, VP of HR at Lamp Electronics is sitting scratching his head over the conversation he had with Joe Group describing what had transpired in the finance department. Joe had been preparing...
-
Give some other examples of public goods. In each case, explain why additional users do not deplete the good and why it is difficult to exclude people from using it.
-
Spensers Stores carried out the following transactions relating to a single product in October. Required (a) Prepare general journal entries to record the transactions, assuming that a perpetual...
-
Microtech Corporation is expanding rapidly and currently needs to retain all of its earnings; hence, it does not pay dividends. However, investors expect Microtech to begin paying dividends,...
-
33. The dimensions of Resistance is (1) LMT-A-] (2) [L-2M- T A] (3) [L M- T A] (4) [L MT A]
-
The Excel file Restaurant Sales provides sample information on lunch, dinner, and delivery sales for a local Italian restaurant. Develop 95% prediction intervals for the daily dollar sales of each of...
-
(a) Write the possible oxidation and reduction half-reactions that occur when magnesium is immersed in each of the following solutions: (i) HCl, (ii) an HCl solution containing dissolved oxygen,...
-
(a) Compute the voltage at 25C of an electrochemical cell consisting of pure cadmium immersed in a 2 ( 10-3 M solution of Cd2+ ions, and pure iron in a 0.4 M solution of Fe2+ ions. (b) Write the...
-
Derive the equations necessary to expand Illustration 6.4-1 to include the thermodynamic state variables internal energy, Gibbs energy, and Helmholtz energy. Illustration 6.4-1 Making of a...
-
Q6. A jet of water from a nozzle is deflected through an angle a=60 from its original direction by a curved vane which it enters tangentially without shock with a mean velocity of 25 m/s. If the...
-
16. Solve for the cos (45).
-
The Berkel Corporation manufactures Widgets, Gizmos, and Turnbols from a joint process. June production is 10,000 widgets; 13,500 gizmos; and 15,000 turnbols. Respective per unit selling prices at...
-
Christopher starts a retirement fund 30 years before retirement. He pays $25 per month into the annuity for 30 years with an interest rate of 5% per year compounded monthly. Find the value of...
-
During the 12 - month reporting period ended 31 December 20X8 Keele decreased its allowance for receivables by E600. An irrecoverable debt written off in the previous reporting period amounting to...
-
The step response of a linear circuit is \(g(t)\) \(=\left[5 e^{-1000 t}ight] u(t)\). Find the sinusoidal steady-state output for an input \(x(t)=10 \cos 1000 t\).
-
A Bloomberg Businessweek subscriber study asked, In the past 12 months, when traveling for business, what type of airline ticket did you purchase most often? A second question asked if the type of...
-
Write a query to display the book number, title, and year of all books published after 2015 and on the Programming subject, sorted by book number (Figure P7.68). Figure P7.68 Newer Books on...
-
The component pictured in Figure CS-16 is the impeller of a water pump used by a major automotive manufacturer. The outer diameter of the component is 2.75 in. and the total height of the six curved...
-
What is plasticity?
-
Why is it important to be able to predict the forces or powers required to perform specific forming processes?
-
Prove that Russian multiplication does what it needs to do, i.e. the result is the product of the two integers. Do not use the proof of the book. It is mainly an exercise in understanding the binary...
-
What is the fundament difference between growing a unicorn versus a camel? What are the financial recommendations for start-ups and the reasons given for those choices? Do you agree with these...
-
16. Nickel Inc. bought $500,000 of 3-year, 9% bonds as an investment on December 31, 2015 for $545,000. Nickel uses straight-line amortization. On May 1, 2016, $100,000 of the bonds were redeemed at...

Study smarter with the SolutionInn App


